Target Information
| Target General Information | Top | |||||
|---|---|---|---|---|---|---|
| Target ID |
T60708
|
|||||
| Target Name |
Complement C5 (CO5)
|
|||||
| Synonyms |
C3 and PZP-like alpha-2-macroglobulin domain-containing protein 4
Click to Show/Hide
|
|||||
| Gene Name |
C5
|
|||||
| Target Type |
Successful target
|
[1] | ||||
| Disease | [+] 5 Target-related Diseases | + | ||||
| 1 | Diffuse large B-cell lymphoma ICD-11: 2A81 | |||||
| 2 | Haemolytic anemia [ICD-11: 3A20-3A2Z] | |||||
| 3 | Macular degeneration ICD-11: 9B75 | |||||
| 4 | Myasthenia gravis ICD-11: 8C60 | |||||
| 5 | Protein-losing enteropathy ICD-11: DA96 | |||||
| Function |
Activation of C5 by a C5 convertase initiates the spontaneous assembly of the late complement components, C5-C9, into the membrane attack complex. C5b has a transient binding site for C6. The C5b-C6 complex is the foundation upon which the lytic complex is assembled. Derived from proteolytic degradation of complement C5, C5 anaphylatoxin is a mediator of local inflammatory process. Binding to the receptor C5AR1 induces a variety of responses including intracellular calcium release, contraction of smooth muscle, increased vascular permeability, and histamine release from mast cells and basophilic leukocytes (PubMed:8182049). C5a is also a potent chemokine which stimulates the locomotion of polymorphonuclear leukocytes and directs their migration toward sites of inflammation.
Click to Show/Hide
|
|||||
| BioChemical Class |
Complement system
|
|||||
| UniProt ID | ||||||
| Sequence |
MGLLGILCFLIFLGKTWGQEQTYVISAPKIFRVGASENIVIQVYGYTEAFDATISIKSYP
DKKFSYSSGHVHLSSENKFQNSAILTIQPKQLPGGQNPVSYVYLEVVSKHFSKSKRMPIT YDNGFLFIHTDKPVYTPDQSVKVRVYSLNDDLKPAKRETVLTFIDPEGSEVDMVEEIDHI GIISFPDFKIPSNPRYGMWTIKAKYKEDFSTTGTAYFEVKEYVLPHFSVSIEPEYNFIGY KNFKNFEITIKARYFYNKVVTEADVYITFGIREDLKDDQKEMMQTAMQNTMLINGIAQVT FDSETAVKELSYYSLEDLNNKYLYIAVTVIESTGGFSEEAEIPGIKYVLSPYKLNLVATP LFLKPGIPYPIKVQVKDSLDQLVGGVPVTLNAQTIDVNQETSDLDPSKSVTRVDDGVASF VLNLPSGVTVLEFNVKTDAPDLPEENQAREGYRAIAYSSLSQSYLYIDWTDNHKALLVGE HLNIIVTPKSPYIDKITHYNYLILSKGKIIHFGTREKFSDASYQSINIPVTQNMVPSSRL LVYYIVTGEQTAELVSDSVWLNIEEKCGNQLQVHLSPDADAYSPGQTVSLNMATGMDSWV ALAAVDSAVYGVQRGAKKPLERVFQFLEKSDLGCGAGGGLNNANVFHLAGLTFLTNANAD DSQENDEPCKEILRPRRTLQKKIEEIAAKYKHSVVKKCCYDGACVNNDETCEQRAARISL GPRCIKAFTECCVVASQLRANISHKDMQLGRLHMKTLLPVSKPEIRSYFPESWLWEVHLV PRRKQLQFALPDSLTTWEIQGVGISNTGICVADTVKAKVFKDVFLEMNIPYSVVRGEQIQ LKGTVYNYRTSGMQFCVKMSAVEGICTSESPVIDHQGTKSSKCVRQKVEGSSSHLVTFTV LPLEIGLHNINFSLETWFGKEILVKTLRVVPEGVKRESYSGVTLDPRGIYGTISRRKEFP YRIPLDLVPKTEIKRILSVKGLLVGEILSAVLSQEGINILTHLPKGSAEAELMSVVPVFY VFHYLETGNHWNIFHSDPLIEKQKLKKKLKEGMLSIMSYRNADYSYSVWKGGSASTWLTA FALRVLGQVNKYVEQNQNSICNSLLWLVENYQLDNGSFKENSQYQPIKLQGTLPVEAREN SLYLTAFTVIGIRKAFDICPLVKIDTALIKADNFLLENTLPAQSTFTLAISAYALSLGDK THPQFRSIVSALKREALVKGNPPIYRFWKDNLQHKDSSVPNTGTARMVETTAYALLTSLN LKDINYVNPVIKWLSEEQRYGGGFYSTQDTINAIEGLTEYSLLVKQLRLSMDIDVSYKHK GALHNYKMTDKNFLGRPVEVLLNDDLIVSTGFGSGLATVHVTTVVHKTSTSEEVCSFYLK IDTQDIEASHYRGYGNSDYKRIVACASYKPSREESSSGSSHAVMDISLPTGISANEEDLK ALVEGVDQLFTDYQIKDGHVILQLNSIPSSDFLCVRFRIFELFEVGFLSPATFTVYEYHR PDKQCTMFYSTSNIKIQKVCEGAACKCVEADCGQMQEELDLTISAETRKQTACKPEIAYA YKVSITSITVENVFVKYKATLLDIYKTGEAVAEKDSEITFIKKVTCTNAELVKGRQYLIM GKEALQIKYNFSFRYIYPLDSLTWIEYWPRDTTCSSCQAFLANLDEFAEDIFLNGC Click to Show/Hide
|
|||||
| 3D Structure | Click to Show 3D Structure of This Target | PDB | ||||
| HIT2.0 ID | T80L9F | |||||
| Drugs and Modes of Action | Top | |||||
|---|---|---|---|---|---|---|
| Approved Drug(s) | [+] 4 Approved Drugs | + | ||||
| 1 | Avacincaptad pegol | Drug Info | Approved | Geographic atrophy | [2] | |
| 2 | Pozelimab | Drug Info | Approved | Large B Cell Lymphoma | [3] | |
| 3 | Ravulizumab | Drug Info | Approved | Paroxysmal nocturnal haemoglobinuria | [1] | |
| 4 | Zilucoplan | Drug Info | Approved | Generalized myasthenia gravis | [4] | |
| Clinical Trial Drug(s) | [+] 14 Clinical Trial Drugs | + | ||||
| 1 | ABP959 | Drug Info | Phase 3 | Paroxysmal nocturnal haemoglobinuria | [5] | |
| 2 | ALXN1720 | Drug Info | Phase 3 | Generalized myasthenia gravis | [6] | |
| 3 | Coversin | Drug Info | Phase 3 | Paroxysmal nocturnal haemoglobinuria | [7] | |
| 4 | Nomacopan | Drug Info | Phase 3 | Bullous pemphigoid | [8] | |
| 5 | SB12 | Drug Info | Phase 3 | Paroxysmal nocturnal haemoglobinuria | [9] | |
| 6 | SKY59 | Drug Info | Phase 3 | Haemolytic uraemic syndrome | [10] | |
| 7 | Zimura | Drug Info | Phase 3 | Macular degeneration | [11] | |
| 8 | IFX-1 | Drug Info | Phase 2 | Hidradenitis suppurativa | [12] | |
| 9 | LFG-316 | Drug Info | Phase 2 | Macular degeneration | [13] | |
| 10 | NNC-0151-0000-0000 | Drug Info | Phase 2 | Rheumatoid arthritis | [14] | |
| 11 | Tesidolumab | Drug Info | Phase 2 | Age-related macular degeneration | [15] | |
| 12 | ARC-1905 | Drug Info | Phase 1 | Macular degeneration | [16] | |
| 13 | MEDI7814 | Drug Info | Phase 1 | Chronic obstructive pulmonary disease | [17] | |
| 14 | REV-576 | Drug Info | Phase 1 | Autoimmune diabetes | [18] | |
| Discontinued Drug(s) | [+] 3 Discontinued Drugs | + | ||||
| 1 | Pexelizumab | Drug Info | Discontinued in Phase 3 | Cardiovascular disease | [19] | |
| 2 | MP-435 | Drug Info | Discontinued in Phase 2 | Rheumatoid arthritis | [20] | |
| 3 | CGS-32359 | Drug Info | Terminated | Inflammation | [22] | |
| Preclinical Drug(s) | [+] 1 Preclinical Drugs | + | ||||
| 1 | SAND-5 | Drug Info | Preclinical | Glomerulonephritis | [21] | |
| Mode of Action | [+] 3 Modes of Action | + | ||||
| Inhibitor | [+] 15 Inhibitor drugs | + | ||||
| 1 | Avacincaptad pegol | Drug Info | [23] | |||
| 2 | Pozelimab | Drug Info | [3] | |||
| 3 | Ravulizumab | Drug Info | [1] | |||
| 4 | Zilucoplan | Drug Info | [4] | |||
| 5 | ABP959 | Drug Info | [24] | |||
| 6 | Coversin | Drug Info | [26] | |||
| 7 | Nomacopan | Drug Info | [27] | |||
| 8 | SB12 | Drug Info | [28] | |||
| 9 | SKY59 | Drug Info | [29] | |||
| 10 | Zimura | Drug Info | [30] | |||
| 11 | IFX-1 | Drug Info | [12] | |||
| 12 | Tesidolumab | Drug Info | [32] | |||
| 13 | REV-576 | Drug Info | [34] | |||
| 14 | SAND-5 | Drug Info | [37] | |||
| 15 | CGS-32359 | Drug Info | [38] | |||
| Modulator | [+] 1 Modulator drugs | + | ||||
| 1 | MEDI7814 | Drug Info | [33] | |||
| Antagonist | [+] 1 Antagonist drugs | + | ||||
| 1 | MP-435 | Drug Info | [36] | |||
| Cell-based Target Expression Variations | Top | |||||
|---|---|---|---|---|---|---|
| Cell-based Target Expression Variations | ||||||
| Drug Binding Sites of Target | Top | |||||
|---|---|---|---|---|---|---|
| Ligand Name: N-(2-Acetamido)Iminodiacetic Acid | Ligand Info | |||||
| Structure Description | Crystal structure of human complement C5 in complex with the K8 bovine knob domain peptide. | PDB:7AD7 | ||||
| Method | X-ray diffraction | Resolution | 2.30 Å | Mutation | No | [39] |
| PDB Sequence |
EQTYVISAPK
29 IFRVGASENI39 VIQVYGYTEA49 FDATISIKSY59 PDKKFSYSSG69 HVHLSSENKF 79 QNSAILTIQP89 KQLPGGQNPV99 SYVYLEVVSK109 HFSKSKRMPI119 TYDNGFLFIH 129 TDKPVYTPDQ139 SVKVRVYSLN149 DDLKPAKRET159 VLTFIDPEGS169 EVDMVEEIDH 179 IGIISFPDFK189 IPSNPRYGMW199 TIKAKYKEDF209 STTGTAYFEV219 KEYVLPHFSV 229 SIEPEYNFIG239 YKNFKNFEIT249 IKARYFYNKV259 VTEADVYITF269 GIREDLKDDQ 279 KEMMQTAMQN289 TMLINGIAQV299 TFDSETAVKE309 LSYYSLEDLN319 NKYLYIAVTV 329 IESTGGFSEE339 AEIPGIKYVL349 SPYKLNLVAT359 PLFLKPGIPY369 PIKVQVKDSL 379 DQLVGGVPVT389 LNAQTIDVNQ399 ETSDLDPSKS409 VTRVDDGVAS419 FVLNLPSGVT 429 VLEFNVKTDA439 PDLPEENQAR449 EGYRAIAYSS459 LSQSYLYIDW469 TDNHKALLVG 479 EHLNIIVTPK489 SPYIDKITHY499 NYLILSKGKI509 IHFGTREKFS519 DASYQSINIP 529 VTQNMVPSSR539 LLVYYIVTGE549 QTAELVSDSV559 WLNIEEKCGN569 QLQVHLSPDA 579 DAYSPGQTVS589 LNMATGMDSW599 VALAAVDSAV609 YGFQFLEKSD631 LGCGAGGGLN 641 NANVFHLAGL651 TFLTNANADD661 SQCKE
|
|||||
|
|
||||||
| Click to View More Binding Site Information of This Target with Different Ligands | ||||||
| Different Human System Profiles of Target | Top |
|---|---|
|
Human Similarity Proteins
of target is determined by comparing the sequence similarity of all human proteins with the target based on BLAST. The similarity proteins for a target are defined as the proteins with E-value < 0.005 and outside the protein families of the target.
A target that has fewer human similarity proteins outside its family is commonly regarded to possess a greater capacity to avoid undesired interactions and thus increase the possibility of finding successful drugs
(Brief Bioinform, 21: 649-662, 2020).
Human Tissue Distribution
of target is determined from a proteomics study that quantified more than 12,000 genes across 32 normal human tissues. Tissue Specificity (TS) score was used to define the enrichment of target across tissues.
The distribution of targets among different tissues or organs need to be taken into consideration when assessing the target druggability, as it is generally accepted that the wider the target distribution, the greater the concern over potential adverse effects
(Nat Rev Drug Discov, 20: 64-81, 2021).
Human Pathway Affiliation
of target is determined by the life-essential pathways provided on KEGG database. The target-affiliated pathways were defined based on the following two criteria (a) the pathways of the studied target should be life-essential for both healthy individuals and patients, and (b) the studied target should occupy an upstream position in the pathways and therefore had the ability to regulate biological function.
Targets involved in a fewer pathways have greater likelihood to be successfully developed, while those associated with more human pathways increase the chance of undesirable interferences with other human processes
(Pharmacol Rev, 58: 259-279, 2006).
Biological Network Descriptors
of target is determined based on a human protein-protein interactions (PPI) network consisting of 9,309 proteins and 52,713 PPIs, which were with a high confidence score of ≥ 0.95 collected from STRING database.
The network properties of targets based on protein-protein interactions (PPIs) have been widely adopted for the assessment of target’s druggability. Proteins with high node degree tend to have a high impact on network function through multiple interactions, while proteins with high betweenness centrality are regarded to be central for communication in interaction networks and regulate the flow of signaling information
(Front Pharmacol, 9, 1245, 2018;
Curr Opin Struct Biol. 44:134-142, 2017).
Human Similarity Proteins
Human Tissue Distribution
Human Pathway Affiliation
Biological Network Descriptors
|
|
|
There is no similarity protein (E value < 0.005) for this target
|
|
Note:
If a protein has TS (tissue specficity) scores at least in one tissue >= 2.5, this protein is called tissue-enriched (including tissue-enriched-but-not-specific and tissue-specific). In the plots, the vertical lines are at thresholds 2.5 and 4.
|

| KEGG Pathway | Pathway ID | Affiliated Target | Pathway Map |
|---|---|---|---|
| Neuroactive ligand-receptor interaction | hsa04080 | Affiliated Target |

|
| Class: Environmental Information Processing => Signaling molecules and interaction | Pathway Hierarchy | ||
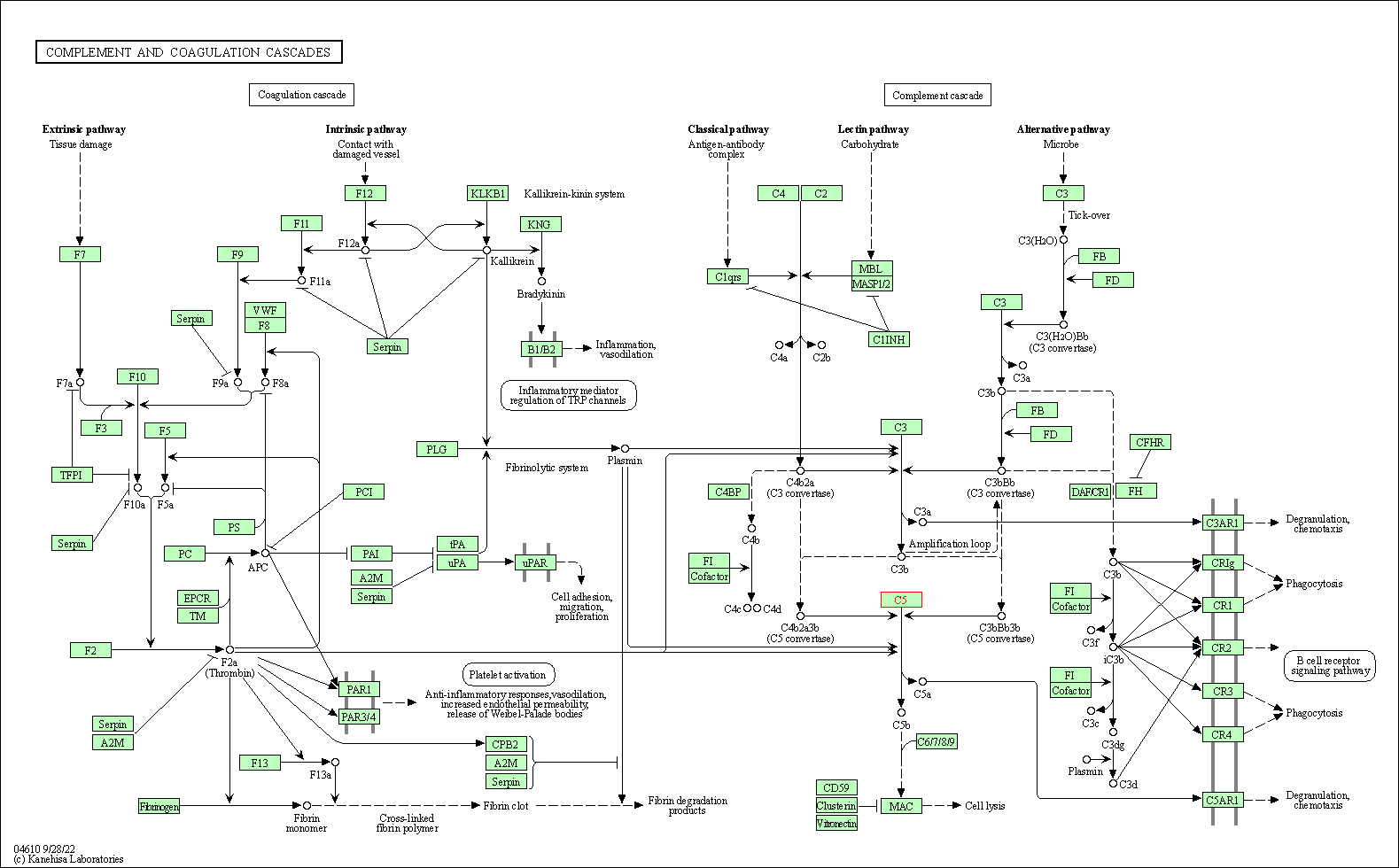
| Complement and coagulation cascades | hsa04610 | Affiliated Target |

|
| Class: Organismal Systems => Immune system | Pathway Hierarchy | ||
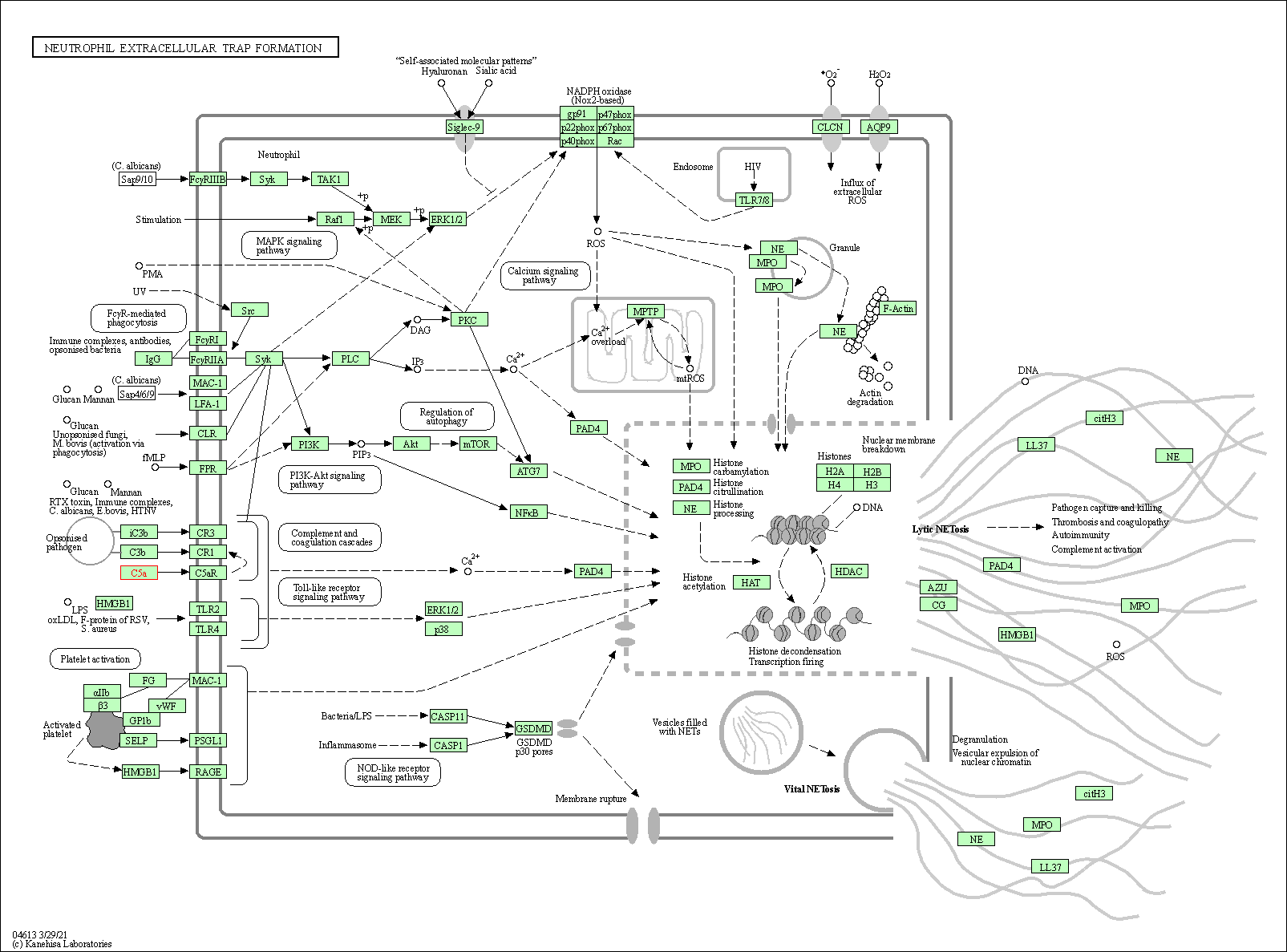
| Neutrophil extracellular trap formation | hsa04613 | Affiliated Target |

|
| Class: Organismal Systems => Immune system | Pathway Hierarchy | ||
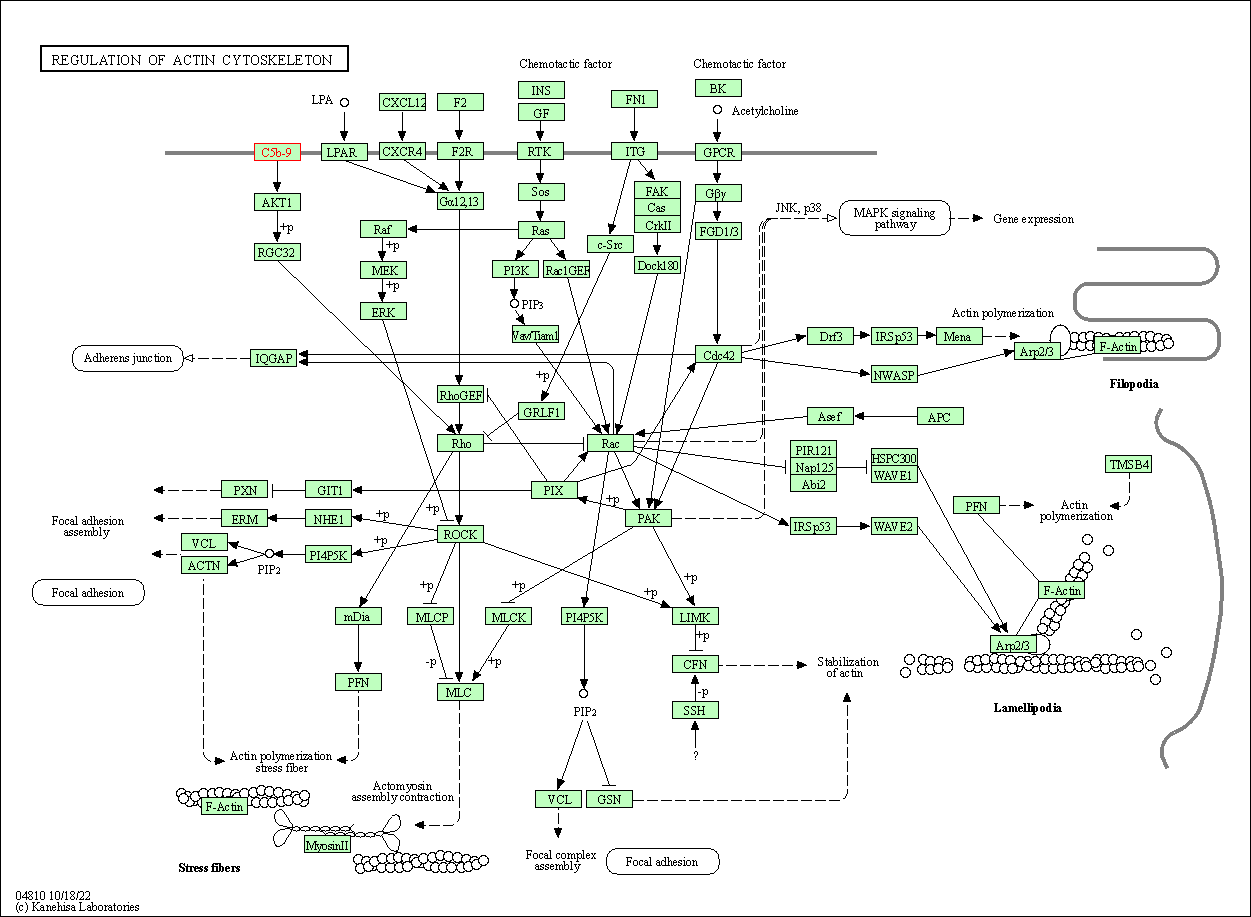
| Regulation of actin cytoskeleton | hsa04810 | Affiliated Target |

|
| Class: Cellular Processes => Cell motility | Pathway Hierarchy | ||
| Degree | 11 | Degree centrality | 1.18E-03 | Betweenness centrality | 8.19E-04 |
|---|---|---|---|---|---|
| Closeness centrality | 1.72E-01 | Radiality | 1.27E+01 | Clustering coefficient | 2.91E-01 |
| Neighborhood connectivity | 5.36E+00 | Topological coefficient | 2.42E-01 | Eccentricity | 14 |
| Download | Click to Download the Full PPI Network of This Target | ||||
| Target Affiliated Biological Pathways | Top | |||||
|---|---|---|---|---|---|---|
| KEGG Pathway | [+] 6 KEGG Pathways | + | ||||
| 1 | Complement and coagulation cascades | |||||
| 2 | Prion diseases | |||||
| 3 | Pertussis | |||||
| 4 | Staphylococcus aureus infection | |||||
| 5 | Herpes simplex infection | |||||
| 6 | Systemic lupus erythematosus | |||||
| Reactome | [+] 4 Reactome Pathways | + | ||||
| 1 | Activation of C3 and C5 | |||||
| 2 | Peptide ligand-binding receptors | |||||
| 3 | G alpha (i) signalling events | |||||
| 4 | Regulation of Complement cascade | |||||
| WikiPathways | [+] 7 WikiPathways | + | ||||
| 1 | Complement Activation, Classical Pathway | |||||
| 2 | Human Complement System | |||||
| 3 | Spinal Cord Injury | |||||
| 4 | Allograft Rejection | |||||
| 5 | GPCR ligand binding | |||||
| 6 | GPCR downstream signaling | |||||
| 7 | Complement cascade | |||||
| References | Top | |||||
|---|---|---|---|---|---|---|
| REF 1 | 2018 FDA drug approvals.Nat Rev Drug Discov. 2019 Feb;18(2):85-89. | |||||
| REF 2 | FDA Approved Drug Products from FDA Official Website. 2023. Application Number: 217225 | |||||
| REF 3 | FDA Approved Drug Products from FDA Official Website. 2023. Application Number: 761339 | |||||
| REF 4 | FDA Approved Drug Products from FDA Official Website. 2023. Application Number: 216834 | |||||
| REF 5 | ClinicalTrials.gov (NCT03818607) A Study Evaluating the Efficacy and Safety of ABP 959 Compared With Eculizumab in Adult Participants With PNH (DAHLIA). U.S. National Institutes of Health. | |||||
| REF 6 | ClinicalTrials.gov (NCT05556096) A Phase 3, Randomized, Double-blind, Placebo-controlled, Parallel, Multicenter Study to Evaluate the Safety and Efficacy of ALXN1720 in Adults With Generalized Myasthenia Gravis. U.S.National Institutes of Health. | |||||
| REF 7 | ClinicalTrials.gov (NCT03427060) Coversin in PNH in Patients With Resistance to Eculizumab Due to Complement C5 Polymorphisms (CONSENTII). U.S. National Institutes of Health. | |||||
| REF 8 | ClinicalTrials.gov (NCT05061771) A Randomized, Part A Partial Blinded and Part B Double Blinded, Placebo-controlled 24-week Clinical Study to Evaluate the Efficacy and Safety of Nomacopan Therapy in Adult Patients With Bullous Pemphigoid Receiving Adjunct Oral Corticosteroid Therapy (ARREST-BP). U.S.National Institutes of Health. | |||||
| REF 9 | ClinicalTrials.gov (NCT04058158) A Study to Compare SB12 (Proposed Eculizumab Biosimilar) to Soliris in Subjects With Paroxysmal Nocturnal Haemoglobinuria. U.S. National Institutes of Health. | |||||
| REF 10 | ClinicalTrials.gov (NCT04861259) A Study Evaluating the Efficacy, Safety, Pharmacokinetics and Pharmacodynamics of Crovalimab in Adult and Adolescent Participants With Atypical Hemolytic Uremic Syndrome (aHUS) (COMMUTE-a). U.S. National Institutes of Health. | |||||
| REF 11 | ClinicalTrials.gov (NCT04435366) A Phase 3 Safety and Efficacy Study of Intravitreal Administration of Zimura (Complement C5 Inhibitor). U.S. National Institutes of Health. | |||||
| REF 12 | Clinical pipeline report, company report or official report of the Pharmaceutical Research and Manufacturers of America (PhRMA) | |||||
| REF 13 | ClinicalTrials.gov (NCT01527500) Intravitreal LFG316 in Patients With Age-related Macular Degeneration (AMD). U.S. National Institutes of Health. | |||||
| REF 14 | Complement-targeted therapeutics. Nat Biotechnol. 2007 Nov;25(11):1265-75. | |||||
| REF 15 | ClinicalTrials.gov (NCT01527500) Intravitreal LFG316 in Patients With Age-related Macular Degeneration (AMD). U.S. National Institutes of Health. | |||||
| REF 16 | ClinicalTrials.gov (NCT00950638) A Study of ARC1905 (Anti-C5 Aptamer) in Subjects With Dry Age-related Macular Degeneration. U.S. National Institutes of Health. | |||||
| REF 17 | ClinicalTrials.gov (NCT01544361) A Phase 1 Study to Evaluate the Safety of MEDI7814 in Adult Volunteers. U.S. National Institutes of Health. | |||||
| REF 18 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800042907) | |||||
| REF 19 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800010238) | |||||
| REF 20 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800027730) | |||||
| REF 21 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800032848) | |||||
| REF 22 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800010131) | |||||
| REF 23 | FDA Approved Drug Products from FDA Official Website. 2023. Application Number: 217225 | |||||
| REF 24 | A randomized, double-blind, single-dose, three-arm, parallel group study to determine pharmacokinetic similarity of ABP 959 and eculizumab (Soliris ?) in healthy male subjects. Eur J Haematol. 2020 Jul;105(1):66-74. | |||||
| REF 25 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2023. Adis Insight | |||||
| REF 26 | Use of the complement inhibitor Coversin to treat HSCT-associated TMA. Blood Adv. 2017 Jul 3;1(16):1254-1258. | |||||
| REF 27 | Evaluation of Nomacopan for Treatment of Bullous Pemphigoid: A Phase 2a Nonrandomized Controlled Trial. JAMA Dermatol. 2022 Jun 1;158(6):641-649. | |||||
| REF 28 | Clinical promise of next-generation complement therapeutics. Nat Rev Drug Discov. 2019 Sep;18(9):707-729. | |||||
| REF 29 | The complement C5 inhibitor crovalimab in paroxysmal nocturnal hemoglobinuria. Blood. 2020 Mar 19;135(12):912-920. | |||||
| REF 30 | C5 Inhibitor Avacincaptad Pegol for Geographic Atrophy Due to Age-Related Macular Degeneration: A Randomized Pivotal Phase 2/3 Trial. Ophthalmology. 2021 Apr;128(4):576-586. | |||||
| REF 31 | Advances in the management of macular degeneration. F1000Prime Rep. 2014; 6: 29. | |||||
| REF 32 | Intravenous immunoglobulin significantly reduces exposure of concomitantly administered anti-C5 monoclonal antibody tesidolumab. Am J Transplant. 2020 Sep;20(9):2581-2588. | |||||
| REF 33 | Interpreting expression profiles of cancers by genome-wide survey of breadth of expression in normal tissues. Genomics 2005 Aug;86(2):127-41. | |||||
| REF 34 | Complement C5-inhibitor rEV576 (coversin) ameliorates in-vivo effects of antiphospholipid antibodies. Lupus. 2014 Oct;23(12):1324-6. | |||||
| REF 35 | Pexelizumab reduces death and myocardial infarction in higher risk cardiac surgical patients. Ann Thorac Surg. 2006 Aug;82(2):486-92. | |||||
| REF 36 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800027730) | |||||
| REF 37 | Progress and Trends in Complement Therapeutics. Adv Exp Med Biol. 2013; 734: 1-22. | |||||
| REF 38 | Anticomplement therapy. Biologics. 2008 December; 2(4): 671-685. | |||||
| REF 39 | The allosteric modulation of complement C5 by knob domain peptides. Elife. 2021 Feb 11;10:e63586. | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

