| Drug General Information |
| Drug ID |
D0D9FR
|
| Former ID |
DNC004712
|
| Drug Name |
4-[5-Bromo-indan-(1Z)-ylidenemethyl]-pyridine
|
| Drug Type |
Small molecular drug
|
| Indication |
Discovery agent
|
Investigative |
[1]
|
|---|
| Structure |
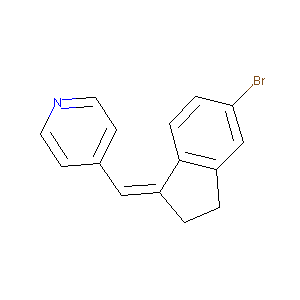
|
Download
2D MOL
3D MOL
|
| Formula |
C15H12BrN
|
| Canonical SMILES |
C1CC(=CC2=CC=NC=C2)C3=C1C=C(C=C3)Br
|
| InChI |
1S/C15H12BrN/c16-14-3-4-15-12(1-2-13(15)10-14)9-11-5-7-17-8-6-11/h3-10H,1-2H2/b12-9-
|
| InChIKey |
MNQUPNOEJZVKRD-XFXZXTDPSA-N
|
| PubChem Compound ID |
|
| Target and Pathway |
| Target(s) |
Cytochrome P450 19 |
Target Info |
Inhibitor |
[1]
|
|---|
| Cytochrome P450 11B1, mitochondrial |
Target Info |
Inhibitor |
[1]
|
| 17 alpha-hydroxylase-C17, 20-lyase |
Target Info |
Inhibitor |
[1]
|
|
BioCyc Pathway
|
Superpathway of steroid hormone biosynthesis
|
|
Estradiol biosynthesis II
|
|
Estradiol biosynthesis IPWY-7305:Superpathway of steroid hormone biosynthesis
|
|
Glucocorticoid biosynthesis
|
|
Mineralocorticoid biosynthesisPWY-7305:Superpathway of steroid hormone biosynthesis
|
|
Androgen biosynthesis
|
|
KEGG Pathway
|
Steroid hormone biosynthesis
|
|
Metabolic pathways
|
|
Ovarian steroidogenesishsa00140:Steroid hormone biosynthesis
|
|
Metabolic pathwayshsa00140:Steroid hormone biosynthesis
|
|
Ovarian steroidogenesis
|
|
Prolactin signaling pathway
|
|
NetPath Pathway
|
FSH Signaling Pathway
|
|
PANTHER Pathway
|
Androgen/estrogene/progesterone biosynthesis
|
|
PathWhiz Pathway
|
Androgen and Estrogen MetabolismPW000141:SteroidogenesisPW000045:Androgen and Estrogen Metabolism
|
|
Steroidogenesis
|
|
Reactome
|
Endogenous sterolsR-HSA-194002:Glucocorticoid biosynthesis
|
|
Endogenous sterolsR-HSA-193048:Androgen biosynthesis
|
|
Glucocorticoid biosynthesis
|
|
Endogenous sterols
|
|
WikiPathways
|
Metapathway biotransformation
|
|
Tryptophan metabolism
|
|
Oxidation by Cytochrome P450
|
|
Ovarian Infertility Genes
|
|
Metabolism of steroid hormones and vitamin D
|
|
FSH signaling pathway
|
|
Integrated Breast Cancer Pathway
|
|
Phase 1 - Functionalization of compoundsWP702:Metapathway biotransformation
|
|
Corticotropin-releasing hormoneWP702:Metapathway biotransformation
|
|
Steroid Biosynthesis
|
|
Glucocorticoid & Mineralcorticoid Metabolism
|
|
Prostate Cancer
|
|
Phase 1 - Functionalization of compounds
|
| References |
| REF 1 | J Med Chem. 2005 Mar 10;48(5):1563-75.Synthesis and evaluation of (pyridylmethylene)tetrahydronaphthalenes/-indanes and structurally modified derivatives: potent and selective inhibitors of aldosterone synthase. |
|---|