| Drug General Information |
| Drug ID |
D0E7FI
|
| Former ID |
DNC004895
|
| Drug Name |
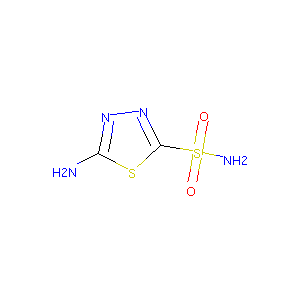
5-amino-1,3,4-thiadiazole-2-sulfonamide
|
| Drug Type |
Small molecular drug
|
| Indication |
Discovery agent
|
Investigative |
[1]
|
|---|
| Structure |
|
Download
2D MOL
3D MOL
|
| Formula |
C2H4N4O2S2
|
| Canonical SMILES |
C1(=NN=C(S1)S(=O)(=O)N)N
|
| InChI |
1S/C2H4N4O2S2/c3-1-5-6-2(9-1)10(4,7)8/h(H2,3,5)(H2,4,7,8)
|
| InChIKey |
VGMVBPQOACUDRU-UHFFFAOYSA-N
|
| PubChem Compound ID |
|
| Target and Pathway |
| Target(s) |
Carbonic anhydrase I |
Target Info |
Inhibitor |
[2]
|
|---|
| Carbonic anhydrase |
Target Info |
Inhibitor |
[1]
|
| Carbonic anhydrase IV |
Target Info |
Inhibitor |
[1]
|
| Carbonic anhydrase XII |
Target Info |
Inhibitor |
[1]
|
| Carbonic anhydrase II |
Target Info |
Inhibitor |
[2]
|
| Carbonic anhydrase XIV |
Target Info |
Inhibitor |
[1]
|
| Carbonic anhydrase IX |
Target Info |
Inhibitor |
[1]
|
| Carbonic anhydrase VI |
Target Info |
Inhibitor |
[1]
|
|
KEGG Pathway
|
Nitrogen metabolismhsa00910:Nitrogen metabolism
|
|
Proximal tubule bicarbonate reclamationhsa00910:Nitrogen metabolismhsa00910:Nitrogen metabolism
|
|
Proximal tubule bicarbonate reclamation
|
|
Collecting duct acid secretion
|
|
Gastric acid secretion
|
|
Pancreatic secretion
|
|
Bile secretionhsa00910:Nitrogen metabolismhsa00910:Nitrogen metabolismhsa00910:Nitrogen metabolism
|
|
NetPath Pathway
|
TGF_beta_Receptor Signaling PathwayNetPath_16:IL4 Signaling Pathway
|
|
EGFR1 Signaling Pathway
|
|
Pathway Interaction Database
|
C-MYB transcription factor networkhif1_tfpathway:HIF-1-alpha transcription factor network
|
|
PathWhiz Pathway
|
Gastric Acid Production
|
|
Reactome
|
Erythrocytes take up carbon dioxide and release oxygen
|
|
Erythrocytes take up oxygen and release carbon dioxide
|
|
Reversible hydration of carbon dioxideR-HSA-1237044:Erythrocytes take up carbon dioxide and release oxygen
|
|
Reversible hydration of carbon dioxideR-HSA-1475029:Reversible hydration of carbon dioxideR-HSA-1237044:Erythrocytes take up carbon dioxide and release oxygen
|
|
Reversible hydration of carbon dioxideR-HSA-1475029:Reversible hydration of carbon dioxideR-HSA-1234158:Regulation of gene expression by Hypoxia-inducible Factor
|
|
Reversible hydration of carbon dioxideR-HSA-1475029:Reversible hydration of carbon dioxide
|
|
WikiPathways
|
Reversible Hydration of Carbon Dioxide
|
|
Uptake of Carbon Dioxide and Release of Oxygen by Erythrocytes
|
|
Uptake of Oxygen and Release of Carbon Dioxide by ErythrocytesWP2770:Reversible Hydration of Carbon Dioxide
|
|
miR-targeted genes in muscle cell - TarBase
|
|
miR-targeted genes in leukocytes - TarBase
|
|
miR-targeted genes in epithelium - TarBaseWP2770:Reversible Hydration of Carbon Dioxide
|
|
Uptake of Oxygen and Release of Carbon Dioxide by ErythrocytesWP2770:Reversible Hydration of Carbon DioxideWP2877:Vitamin D Receptor Pathway
|
|
Regulation of Hypoxia-inducible Factor (HIF) by OxygenWP2770:Reversible Hydration of Carbon Dioxide
|
| References |
| REF 1 | Bioorg Med Chem Lett. 2010 Aug 1;20(15):4376-81. Epub 2010 Jun 17.Carbonic anhydrase inhibitors. The X-ray crystal structure of human isoform II in adduct with an adamantyl analogue of acetazolamideresides in a less utilized binding pocket than most hydrophobic inhibitors. |
|---|
| REF 2 | Eur J Med Chem. 2010 Nov;45(11):4769-73. Epub 2010 Jul 24.Synthesis, characterization and antiglaucoma activity of some novel pyrazole derivatives of 5-amino-1,3,4-thiadiazole-2-sulfonamide. |
|---|