| Drug General Information |
| Drug ID |
D0Y2RL
|
| Former ID |
DNC010002
|
| Drug Name |
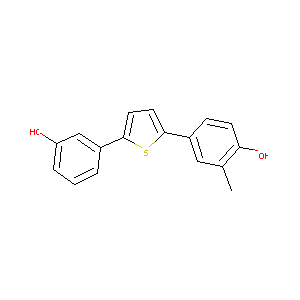
4-[5-(3-Hydroxyphenyl)-2-thienyl)-2-methyl]phenol
|
| Drug Type |
Small molecular drug
|
| Indication |
Discovery agent
|
Investigative |
[1]
|
|---|
| Structure |
|
Download
2D MOL
3D MOL
|
| Formula |
C17H14O2S
|
| Canonical SMILES |
CC1=C(C=CC(=C1)C2=CC=C(S2)C3=CC(=CC=C3)O)O
|
| InChI |
1S/C17H14O2S/c1-11-9-13(5-6-15(11)19)17-8-7-16(20-17)12-3-2-4-14(18)10-12/h2-10,18-19H,1H3
|
| InChIKey |
ICDRVGLMFQWTDX-UHFFFAOYSA-N
|
| PubChem Compound ID |
|
| Target and Pathway |
| Target(s) |
Cytochrome P450 2D6 |
Target Info |
Inhibitor |
[1]
|
|---|
| Estradiol 17 beta-dehydrogenase 1 |
Target Info |
Inhibitor |
[1]
|
| Cytochrome P450 3A4 |
Target Info |
Inhibitor |
[1]
|
|
BioCyc Pathway
|
Superpathway of steroid hormone biosynthesis
|
|
Estradiol biosynthesis I
|
|
KEGG Pathway
|
Metabolism of xenobiotics by cytochrome P450
|
|
Drug metabolism - cytochrome P450
|
|
Serotonergic synapsehsa00140:Steroid hormone biosynthesis
|
|
Metabolic pathways
|
|
Ovarian steroidogenesishsa00140:Steroid hormone biosynthesis
|
|
Linoleic acid metabolism
|
|
Retinol metabolism
|
|
Drug metabolism - other enzymes
|
|
Bile secretion
|
|
Chemical carcinogenesis
|
|
NetPath Pathway
|
FSH Signaling Pathway
|
|
PANTHER Pathway
|
Androgen/estrogene/progesterone biosynthesis
|
|
PathWhiz Pathway
|
Androgen and Estrogen MetabolismPW000015:Caffeine Metabolism
|
|
Retinol Metabolism
|
|
Reactome
|
XenobioticsR-HSA-2453902:The canonical retinoid cycle in rods (twilight vision)R-HSA-211981:Xenobiotics
|
|
Aflatoxin activation and detoxification
|
|
WikiPathways
|
Metapathway biotransformation
|
|
Tamoxifen metabolism
|
|
Oxidation by Cytochrome P450
|
|
Vitamin D Receptor Pathway
|
|
Aripiprazole Metabolic Pathway
|
|
Fatty Acid Omega Oxidation
|
|
Codeine and Morphine MetabolismWP496:Steroid Biosynthesis
|
|
Metabolism of steroid hormones and vitamin D
|
|
Prostate CancerWP702:Metapathway biotransformation
|
|
Aflatoxin B1 metabolism
|
|
Estrogen metabolism
|
|
Benzo(a)pyrene metabolism
|
|
Tryptophan metabolism
|
|
Nuclear Receptors in Lipid Metabolism and Toxicity
|
|
Nuclear Receptors Meta-Pathway
|
|
Farnesoid X Receptor Pathway
|
|
Felbamate Metabolism
|
|
Lidocaine metabolism
|
|
Nifedipine Activity
|
|
Colchicine Metabolic Pathway
|
|
Irinotecan Pathway
|
|
Drug Induction of Bile Acid Pathway
|
|
Codeine and Morphine Metabolism
|
| References |
| REF 1 | J Med Chem. 2009 Nov 12;52(21):6724-43.New insights into the SAR and binding modes of bis(hydroxyphenyl)thiophenes and -benzenes: influence of additional substituents on 17beta-hydroxysteroid dehydrogenase type 1 (17beta-HSD1) inhibitory activity and selectivity. |
|---|