| Drug General Information |
| Drug ID |
D0LC0F
|
| Former ID |
DNC005129
|
| Drug Name |
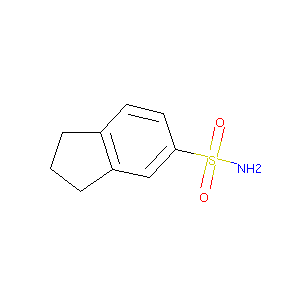
2,3-dihydro-1H-indene-5-sulfonamide
|
| Synonyms |
Indan-5-sulfonic acid amide
|
| Drug Type |
Small molecular drug
|
| Indication |
Discovery agent
|
Investigative |
[1]
|
|---|
| Structure |
|
Download
2D MOL
3D MOL
|
| Formula |
C9H11NO2S
|
| Canonical SMILES |
C1CC2=C(C1)C=C(C=C2)S(=O)(=O)N
|
| InChI |
1S/C9H11NO2S/c10-13(11,12)9-5-4-7-2-1-3-8(7)6-9/h4-6H,1-3H2,(H2,10,11,12)
|
| InChIKey |
XVQJTFMKKZBBSX-UHFFFAOYSA-N
|
| PubChem Compound ID |
|
| Target and Pathway |
| Target(s) |
Carbonic anhydrase |
Target Info |
Inhibitor |
[1]
|
|---|
| Carbonic anhydrase I |
Target Info |
Inhibitor |
[2]
|
| Carbonic anhydrase XIV |
Target Info |
Inhibitor |
[1]
|
| Carbonic anhydrase II |
Target Info |
Inhibitor |
[2]
|
| Carbonic anhydrase XII |
Target Info |
Inhibitor |
[1]
|
|
KEGG Pathway
|
Nitrogen metabolismhsa00910:Nitrogen metabolismhsa00910:Nitrogen metabolism
|
|
Proximal tubule bicarbonate reclamation
|
|
Collecting duct acid secretion
|
|
Gastric acid secretion
|
|
Pancreatic secretion
|
|
Bile secretionhsa00910:Nitrogen metabolism
|
|
NetPath Pathway
|
IL4 Signaling Pathway
|
|
EGFR1 Signaling PathwayNetPath_7:TGF_beta_Receptor Signaling Pathway
|
|
Pathway Interaction Database
|
C-MYB transcription factor network
|
|
PathWhiz Pathway
|
Gastric Acid Production
|
|
Reactome
|
Erythrocytes take up carbon dioxide and release oxygen
|
|
Erythrocytes take up oxygen and release carbon dioxide
|
|
Reversible hydration of carbon dioxideR-HSA-1475029:Reversible hydration of carbon dioxideR-HSA-1237044:Erythrocytes take up carbon dioxide and release oxygen
|
|
Reversible hydration of carbon dioxideR-HSA-1475029:Reversible hydration of carbon dioxide
|
|
WikiPathways
|
Reversible Hydration of Carbon Dioxide
|
|
Uptake of Carbon Dioxide and Release of Oxygen by Erythrocytes
|
|
Uptake of Oxygen and Release of Carbon Dioxide by ErythrocytesWP2770:Reversible Hydration of Carbon DioxideWP2770:Reversible Hydration of Carbon Dioxide
|
|
Uptake of Oxygen and Release of Carbon Dioxide by ErythrocytesWP2770:Reversible Hydration of Carbon Dioxide
|
|
miR-targeted genes in muscle cell - TarBase
|
|
miR-targeted genes in leukocytes - TarBase
|
|
miR-targeted genes in epithelium - TarBase
|
| References |
| REF 1 | Eur J Med Chem. 2008 Dec;43(12):2853-60. Epub 2008 Feb 29.Indanesulfonamides as carbonic anhydrase inhibitors and anticonvulsant agents: structure-activity relationship and pharmacological evaluation. |
|---|
| REF 2 | Bioorg Med Chem Lett. 2004 Dec 6;14(23):5781-6.Carbonic anhydrase inhibitors. Design of anticonvulsant sulfonamides incorporating indane moieties. |
|---|