Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0M0PY
|
||||
| Former ID |
DPR000039
|
||||
| Drug Name |
DX-890
|
||||
| Indication | Acute lung injury; Acute respiratory distress syndrome; Cystic fibrosis [ICD9: 277.0, 518.5, 518.82, 709.2; ICD10:E84, J80, L90.5] | Phase 2 | [1] | ||
| Company |
Dyax Corp.
|
||||
| Structure |
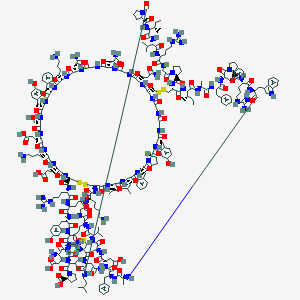
|
Download2D MOL |
|||
| CAS Number |
CAS 10024-97-2
|
||||
| ChEBI ID |
ChEBI:17045
|
||||
| SuperDrug ATC ID |
N01AX13
|
||||
| SuperDrug CAS ID |
cas=010024972
|
||||
| Target and Pathway | |||||
| Target(s) | Leukocyte elastase | Target Info | Inhibitor | [2] | |
| KEGG Pathway | Transcriptional misregulation in cancer | ||||
| Systemic lupus erythematosus | |||||
| Pathway Interaction Database | Urokinase-type plasminogen activator (uPA) and uPAR-mediated signaling | ||||
| C-MYB transcription factor network | |||||
| Reactome | Collagen degradation | ||||
| Degradation of the extracellular matrix | |||||
| Activation of Matrix Metalloproteinases | |||||
| WikiPathways | Hair Follicle Development: Cytodifferentiation (Part 3 of 3) | ||||
| Human Complement System | |||||
| Degradation of collagen | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT00455767) Safety and Efficacy Study of Depelestat in Acute Respiratory Distress Syndrome (ARDS) Patients. U.S. National Institutes of Health. | ||||
| REF 2 | Emerging therapies for treatment of acute lung injury and acute respiratory distress syndrome. Expert Opin Emerg Drugs. 2007 Sep;12(3):461-77. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

