Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0V0SL
|
||||
| Former ID |
DNCL002505
|
||||
| Drug Name |
Abexinostat
|
||||
| Drug Type |
Small molecular drug
|
||||
| Company |
Pharmacyclics
|
||||
| Structure |
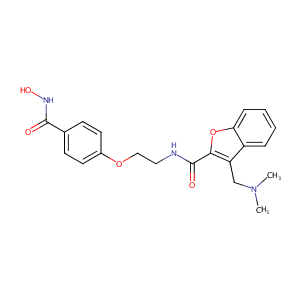
|
Download2D MOL |
|||
| Formula |
C21H23N3O5
|
||||
| InChI |
InChI=1S/C21H23N3O5/c1-24(2)13-17-16-5-3-4-6-18(16)29-19(17)21(26)22-11-12-28-15-9-7-14(8-10-15)20(25)23-27/h3-10,27H,11-13H2,1-2H3,(H,22,26)(H,23,25)
|
||||
| InChIKey |
MAUCONCHVWBMHK-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
16857486, 23832872, 42791900, 56269573, 80326339, 117695997, 124756990, 125163795, 131480753, 134223063, 134964420, 135267137, 135626780, 136340216, 136367289, 136367841, 136920360, 137216456, 139896178, 144116341, 152258290, 152344189, 160647129, 160681721, 162011829, 162037426, 162202783, 163907942, 164041979, 164193977, 172914354, 174006344, 174530736, 175426351, 177748391, 186014504, 198941643, 201507997, 223381303, 223705150, 224769568, 226766445, 248596262, 252166578, 252215998, 252220173, 252671930
|
||||
| Target and Pathway | |||||
| Target(s) | Histone deacetylase | Target Info | Modulator | [533112] | |
| References | |||||
| Ref 543092 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 8366). | ||||
| Ref 548180 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800022814) | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

