Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0E9WL
|
||||
| Former ID |
DAP000434
|
||||
| Drug Name |
Loracarbef
|
||||
| Synonyms |
Lorabid; Loracarbefum; Lorafem; Loracarbef hydrate; KT-3777; LY-163892; Lorabid (TN); (6R,7S)-7-[[(2R)-2-amino-2-phenylacetyl]amino]-3-chloro-8-oxo-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7S)-7-[[(2R)-2-amino-2-phenylacetyl]amino]-3-chloro-8-oxo-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid hydrate; (6R,7S)-7-{[(2R)-2-amino-2-phenylacetyl]amino}-3-chloro-8-oxo-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate; (6R,7S)-7-{[(2R)-2-amino-2-phenylacetyl]amino}-3-chloro-8-oxo-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid; (6R,7S)-7-{[(2R)-2-amino-2-phenylacetyl]amino}-3-chloro-8-oxo-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid hydrate; (6R,7S)-7-{[(2R)-2-ammonio-2-phenylacetyl]amino}-3-chloro-8-oxo-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate; 7beta-[(2R)-2-amino-2-phenylacetyl]nitrilo-3-chloro-3,4-didehydrocepham-4-carboxylate; 7beta-[(2R)-2-amino-2-phenylacetyl]nitrilo-3-chloro-3,4-didehydrocepham-4-carboxylic acid; 7beta-[(2R)-2-ammonio-2-phenylacetyl]nitrilo-3-chloro-3,4-didehydrocepham-4-carboxylate
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Bacterial infections [ICD9: 001-009, 010-018, 020-027, 030-041, 080-088, 090-099, 100-104; ICD10:A00-B99] | Approved | [536361] | ||
| Therapeutic Class |
Antibiotics
|
||||
| Company |
Eli Lilly
|
||||
| Structure |
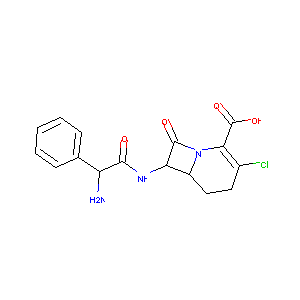
|
Download2D MOL |
|||
| Formula |
C16H16ClN3O4
|
||||
| Canonical SMILES |
C1CC(=C(N2C1C(C2=O)NC(=O)C(C3=CC=CC=C3)N)C(=O)O)Cl
|
||||
| InChI |
1S/C16H16ClN3O4/c17-9-6-7-10-12(15(22)20(10)13(9)16(23)24)19-14(21)11(18)8-4-2-1-3-5-8/h1-5,10-12H,6-7,18H2,(H,19,21)(H,23,24)/t10-,11-,12+/m1/s1
|
||||
| InChIKey |
JAPHQRWPEGVNBT-UTUOFQBUSA-N
|
||||
| CAS Number |
CAS 76470-66-1
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| SuperDrug ATC ID |
J01DC08
|
||||
| SuperDrug CAS ID |
cas=076470661
|
||||
| Target and Pathway | |||||
| Target(s) | DNA | Target Info | Binder | [536267] | |
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

