Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D00FHV
|
||||
| Former ID |
DNC004391
|
||||
| Drug Name |
Iva-His-Pro-Phe-His-ACHPA-Leu-Phe-NH2
|
||||
| Indication | Discovery agent | Investigative | [533493] | ||
| Structure |
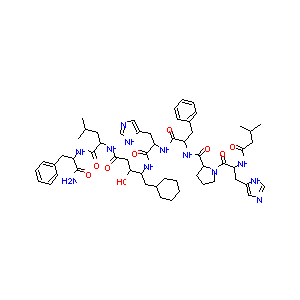
|
Download2D MOL |
|||
| Formula |
C57H80N12O9
|
||||
| Canonical SMILES |
CC(C)CC(C(=O)NC(CC1=CC=CC=C1)C(=O)N)NC(=O)CC(C(CC2CCCCC<br />2)NC(=O)C(CC3=CN=CN3)NC(=O)C(CC4=CC=CC=C4)NC(=O)C5CCCN5<br />C(=O)C(CC6=CN=CN6)NC(=O)CC(C)C)O
|
||||
| InChI |
1S/C57H80N12O9/c1-35(2)23-44(53(74)66-43(52(58)73)26-38-17-10-6-11-18-38)63-51(72)30-49(70)42(25-37-15-8-5-9-16-37)65-55(76)46(28-40-31-59-33-61-40)67-54(75)45(27-39-19-12-7-13-20-39)68-56(77)48-21-14-22-69(48)57(78)47(29-41-32-60-34-62-41)64-50(71)24-36(3)4/h6-7,10-13,17-20,31-37,42-49,70H,5,8-9,14-16,21-30H2,1-4H3,(H2,58,73)(H,59,61)(H,60,62)(H,63,72)(H,64,71)(H,65,76)(H,66,74)(H,67,75)(H,68,77)
|
||||
| InChIKey |
ZOAVSRHEYNKAIW-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Renin, renal | Target Info | Inhibitor | [533493] | |
| KEGG Pathway | Renin-angiotensin system | ||||
| PathWhiz Pathway | Angiotensin Metabolism | ||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

