Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0CW1P
|
||||
| Former ID |
DAP001043
|
||||
| Drug Name |
Betamethasone
|
||||
| Synonyms |
Bebate; Becort; Bedifos; Betacorlan; Betacortril; Betadexamethasone; Betafluorene; Betamamallet; Betametasona; Betametasone; Betamethasonum; Betamethazone; Betapredol; Betasolon; Betnelan; Betsolan; Celestene; Celeston; Celestona; Celestone; Cellestoderm; Cidoten; Flubenisolone; Hormezon; Luxiq; Luxiqo; Methazon; Rinderon; Valisone; Visubeta; Betametasone [DCIT]; Betamethasone Base; Betamethasone Valearate; Betamethasone alcohol; Betamethasone cream; Betamethasonvalerat Mikron; Celestone Syrup and Tablets; Rinderon A; LT00441022; SCH 4831; Beta-Methasone; Beta-Methasone alcohol; Betametasona [INN-Spanish]; Betamethasonum [INN-Latin]; Betnovate (TN); Celestone (TN); Desacort-Beta; Diprolene (TN); Diprosone (TN); Lotrisone (TN); Rinderon (TN); SCH-4831; Betamethasone (JP15/USP/INN); Betamethasone [USAN:BAN:INN:JAN]; Betamethasone [USAN:INN:BAN:JAN]; Celestone, Betadexamethasone, Flubenisolone, Sch-4831, NCS-39470, Betamethasone; 1,4-Pregnadiene-3,20-dione-9alpha-fluoro-16 beta-methyl-11 beta,17alpha,21-triol; 16beta-Methyl-1,4-pregnadiene-9alpha-fluoro-11beta,17alpha,21-triol-3,20-dione; 9-Fluoro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dione; 9-Fluoro-11-beta,17,21-trihydroxy-16-beta-methylpregna-1,4-diene-3,20-dione; 9-Fluoro-11.beta.,17,21-trihydroxy-16.beta.-methylpregna-1,4-diene-3,20-dione; 9-Fluoro-11beta,17,21-trihydroxy-16beta-methylpregna-1,4-diene-3,20-dione; 9-Fluoro-16.beta.-methylprednisolone; 9-Fluoro-16beta-methylprednisolone; 9-alpha-Fluoro-16-beta-methylprednisolone; 9.alpha.-Fluoro-16.beta.-methylprednisolone; 9alpha-Fluoro-16 beta-methyl-prednisolone; 9alpha-Fluoro-16beta-methylprednisolone
|
||||
| Drug Type |
Small molecular drug
|
||||
| Therapeutic Class |
Antiinflammatory Agents
|
||||
| Structure |
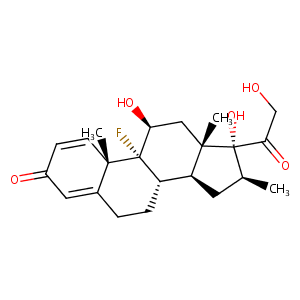
|
Download2D MOL |
|||
| Formula |
C22H29FO5
|
||||
| InChI |
InChI=1S/C22H29FO5/c1-12-8-16-15-5-4-13-9-14(25)6-7-19(13,2)21(15,23)17(26)10-20(16,3)22(12,28)18(27)11-24/h6-7,9,12,15-17,24,26,28H,4-5,8,10-11H2,1-3H3/t12-,15-,16-,17-,19-,20-,21-,22-/m0/s1
|
||||
| InChIKey |
UREBDLICKHMUKA-DVTGEIKXSA-N
|
||||
| CAS Number |
CAS 378-44-9
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
9066, 625879, 3161499, 7847310, 7978781, 8157046, 10321659, 11466390, 11467510, 11486140, 12159278, 14756597, 15004923, 24278276, 24860978, 24892150, 26757820, 29228348, 46500420, 46505155, 46530919, 47193705, 47499670, 47720710, 47720711, 47943900, 48019043, 48415627, 49698457, 50019449, 50111677, 53789445, 56320847, 56462985, 57288752, 57649117, 57653954, 75637045, 85788058, 87564725, 87568418, 92125449, 93167065, 103188847, 103914147, 104253177, 104321493, 121363120, 124757297, 124800522
|
||||
| ChEBI ID |
ChEBI:3077
|
||||
| SuperDrug ATC ID |
A07EA04; C05AA05; D07AC01; D07XC01; H02AB01; R01AD06; R03BA04; S01BA06; S01CB04; S02BA07; S03BA03
|
||||
| SuperDrug CAS ID |
cas=000378449
|
||||
| Target and Pathway | |||||
| Target(s) | Glucocorticoid receptor | Target Info | Agonist | [536246] | |
| KEGG Pathway | Neuroactive ligand-receptor interaction | ||||
| NetPath Pathway | IL2 Signaling Pathway | ||||
| TCR Signaling Pathway | |||||
| References | |||||
| Ref 538521 | FDA Approved Drug Products from FDA Official Website. 2009. Application Number: (NDA) 019137. | ||||
| Ref 542068 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7061). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

