Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0TL7L
|
||||
| Former ID |
DNC009002
|
||||
| Drug Name |
Dexloxiglumide
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Pancreatic disease [ICD10:K85-K86] | Phase 2 | [1] | ||
| Structure |
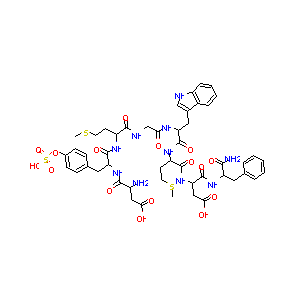
|
Download2D MOL |
|||
| Formula |
C21H30Cl2N2O5
|
||||
| Canonical SMILES |
CSCCC(C(=O)NCC(=O)NC(CC1=CNC2=CC=CC=C21)C(=O)NC(CCSC)C(<br />=O)NC(CC(=O)O)C(=O)NC(CC3=CC=CC=C3)C(=O)N)NC(=O)C(CC4=C<br />C=C(C=C4)OS(=O)(=O)O)NC(=O)C(CC(=O)O)N
|
||||
| InChI |
1S/C49H62N10O16S3/c1-76-18-16-34(55-47(69)37(58-44(66)32(50)23-41(61)62)21-28-12-14-30(15-13-28)75-78(72,73)74)45(67)53-26-40(60)54-38(22-29-25-52-33-11-7-6-10-31(29)33)48(70)56-35(17-19-77-2)46(68)59-39(24-42(63)64)49(71)57-36(43(51)65)20-27-8-4-3-5-9-27/h3-15,25,32,34-39,52H,16-24,26,50H2,1-2H3,(H2,51,65)(H,53,67)(H,54,60)(H,55,69)(H,56,70)(H,57,71)(H,58,66)(H,59,68)(H,61,62)(H,63,64)(H,72,73,74)
|
||||
| InChIKey |
IZTQOLKUZKXIRV-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 25126-32-3
|
||||
| PubChem Compound ID | |||||
| SuperDrug ATC ID |
V04CC03; V04CK02
|
||||
| Target and Pathway | |||||
| Target(s) | Gastrin/cholecystokinin type B receptor | Target Info | Inhibitor | [2] | |
| Cholecystokinin receptor type A | Target Info | Inhibitor | [3] | ||
| KEGG Pathway | Calcium signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Gastric acid secretionhsa04020:Calcium signaling pathway | |||||
| Insulin secretion | |||||
| Pancreatic secretion | |||||
| PANTHER Pathway | CCKR signaling map ST | ||||
| PathWhiz Pathway | Gastric Acid Production | ||||
| Reactome | Peptide ligand-binding receptors | ||||
| G alpha (q) signalling events | |||||
| Gastrin-CREB signalling pathway via PKC and MAPKR-HSA-375276:Peptide ligand-binding receptors | |||||
| WikiPathways | GPCRs, Class A Rhodopsin-like | ||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| Secretion of Hydrochloric Acid in Parietal Cells | |||||
| Peptide GPCRs | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| GPCRs, OtherWP455:GPCRs, Class A Rhodopsin-like | |||||
| References | |||||
| REF 1 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
| REF 2 | J Med Chem. 1997 Oct 10;40(21):3402-7.Synthesis and stereochemical structure-activity relationships of 1,3-dioxoperhydropyrido[1,2-c]pyrimidine derivatives: potent and selective cholecystokinin-A receptor antagonists. | ||||
| REF 3 | J Med Chem. 1989 Feb;32(2):445-9.Full agonists of CCK8 containing a nonhydrolyzable sulfated tyrosine residue. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

