Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0ST5P
|
||||
| Former ID |
DIB000411
|
||||
| Drug Name |
TA-5538
|
||||
| Synonyms |
IDDBCP232835
|
||||
| Indication | Overactive bladder disorder [ICD9: 188, 596.51; ICD10:C67, N32.81] | Discontinued in Phase 2 | [1] | ||
| Company |
Tanabe Seiyaku Co Ltd
|
||||
| Structure |
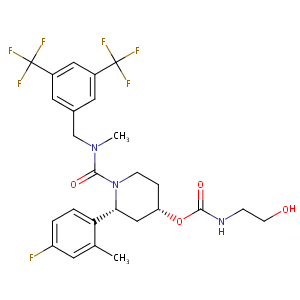
|
Download2D MOL |
|||
| Canonical SMILES |
N1([C@H](C[C@H](CC1)OC(=O)NCCO)c1c(cc(cc1)F)C)C(=O)N(C)<br />Cc1cc(cc(c1)C(F)(F)F)C(F)(F)F
|
||||
| Target and Pathway | |||||
| Target(s) | Substance-P receptor | Target Info | Antagonist | [2] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Measles | |||||
| PANTHER Pathway | CCKR signaling map ST | ||||
| Reactome | G alpha (q) signalling events | ||||
| WikiPathways | SIDS Susceptibility Pathways | ||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| Spinal Cord Injury | |||||
| Peptide GPCRs | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800021639) | ||||
| REF 2 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800021639) | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

