Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0S4JK
|
||||
| Former ID |
DCL000170
|
||||
| Drug Name |
MLN8237
|
||||
| Synonyms |
Alisertib
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Cancer [ICD9: 140-229; ICD10:C00-C96] | Phase 3 | [1], [2] | ||
| Therapeutic Class |
Anticancer Agents
|
||||
| Structure |
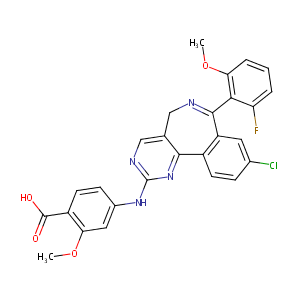
|
Download2D MOL |
|||
| Formula |
C27H20ClFN4O4
|
||||
| InChI |
InChI=1S/C27H20ClFN4O4/c1-36-21-5-3-4-20(29)23(21)25-19-10-15(28)6-8-17(19)24-14(12-30-25)13-31-27(33-24)32-16-7-9-18(26(34)35)22(11-16)37-2/h3-11,13H,12H2,1-2H3,(H,34,35)(H,31,32,33)
|
||||
| InChIKey |
ZLHFILGSQDJULK-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
49693830, 57107096, 99436914, 103651972, 103905632, 104120110, 123055410, 124757022, 125163826, 131477692, 131480698, 134223405, 134338845, 135263892, 135626804, 135727405, 136367323, 136920350, 137173580, 137276087, 141669651, 143499693, 144116244, 152064066, 152234949, 152258281, 152344273, 160647120, 160838255, 162011658, 162037455, 162202572, 163098799, 163403893, 164193986, 164339418, 172095365, 172918135, 174006392, 174525840, 180111921, 185998541, 188899555, 198972240, 198993425, 204398557, 223366121, 223382037, 223613049, 223705104
|
||||
| Target and Pathway | |||||
| Target(s) | Aurora kinase A | Target Info | Inhibitor | [3] | |
| KEGG Pathway | Oocyte meiosis | ||||
| Pathway Interaction Database | Aurora B signaling | ||||
| Signaling by Aurora kinases | |||||
| Integrin-linked kinase signaling | |||||
| PLK1 signaling events | |||||
| Aurora A signaling | |||||
| Reactome | APC/C:Cdh1 mediated degradation of Cdc20 and other APC/C:Cdh1 targeted proteins in late mitosis/early G1 | ||||
| Regulation of PLK1 Activity at G2/M Transition | |||||
| WikiPathways | EGF/EGFR Signaling Pathway | ||||
| JAK/STAT | |||||
| Gastric Cancer Network 1 | |||||
| Integrated Breast Cancer Pathway | |||||
| APC/C-mediated degradation of cell cycle proteins | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT01482962) Alisertib (MLN8237) or Investigator's Choice in Patients With Relapsed/Refractory Peripheral T-Cell Lymphoma. U.S. National Institutes of Health. | ||||
| REF 2 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7790). | ||||
| REF 3 | Effect of Aurora A kinase inhibitor MLN8237 combined with rituximab on antitumor activity in preclinical B-cell non-Hodgkin's lymphoma models. Journal of Clinical Oncology, 2009:8553. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

