Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0P3ZN
|
||||
| Former ID |
DNCL003385
|
||||
| Drug Name |
RDEA3170
|
||||
| Indication | Hyperuricaemia [ICD9: 274, 274.00274.1274.8274.9, 790.6; ICD10:E79.0, M10] | Phase 2 | [524664] | ||
| Company |
AstraZeneca
|
||||
| Structure |
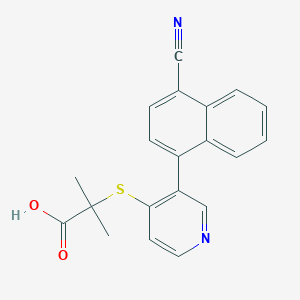
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | Solute carrier family 22 member 12 | Target Info | Modulator | [544332] | |
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

