Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0L7TA
|
||||
| Former ID |
DIB013226
|
||||
| Drug Name |
MIN-101
|
||||
| Indication | Schizophrenia [ICD9: 295; ICD10:F20] | Phase 1 | [1] | ||
| Company |
Minerva neurosciences
|
||||
| Structure |
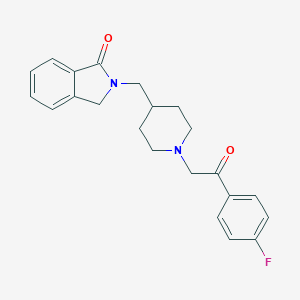
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | 5-hydroxytryptamine 2A receptor | Target Info | Antagonist | [2] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Gap junction | |||||
| Serotonergic synapse | |||||
| Inflammatory mediator regulation of TRP channels | |||||
| PANTHER Pathway | 5HT2 type receptor mediated signaling pathway | ||||
| Reactome | Serotonin receptors | ||||
| G alpha (q) signalling events | |||||
| WikiPathways | Serotonin Receptor 2 and STAT3 Signaling | ||||
| Serotonin Receptor 2 and ELK-SRF/GATA4 signaling | |||||
| SIDS Susceptibility Pathways | |||||
| Monoamine GPCRs | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| GPCRs, Other | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT02232529) Pharmacokinetic Study of MIN-101 in Healthy Subjects. U.S. National Institutes of Health. | ||||
| REF 2 | Pharmacological profile of the new potent neuroleptic ocaperidone (R 79,598). J Pharmacol Exp Ther. 1992 Jan;260(1):146-59. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

