Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0L5WA
|
||||
| Former ID |
DIB012152
|
||||
| Drug Name |
SR-46559A
|
||||
| Synonyms |
6-[2-(Diethylamino)-2-methylpropylamino]-3-phenyl-4-propylpyridazine sesquifumarate
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Cognitive disorders [ICD9: 290-294, 294.0, 780.09, 780.9, 780.93; ICD10:F01-F07, F04, F05, R41.3] | Phase 2 | [1] | ||
| Structure |
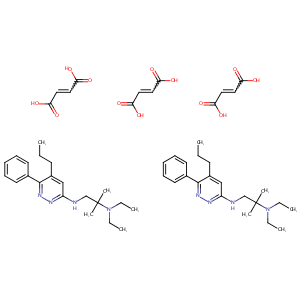
|
Download2D MOL |
|||
| Formula |
C54H76N8O12
|
||||
| Canonical SMILES |
CCCC1=CC(=NN=C1C2=CC=CC=C2)NCC(C)(C)N(CC)CC.CCCC1=CC(=N<br />N=C1C2=CC=CC=C2)NCC(C)(C)N(CC)CC.C(=CC(=O)O)C(=O)O.C(=C<br />C(=O)O)C(=O)O.C(=CC(=O)O)C(=O)O
|
||||
| InChI |
1S/2C21H32N4.3C4H4O4/c2*1-6-12-18-15-19(22-16-21(4,5)25(7-2)8-3)23-24-20(18)17-13-10-9-11-14-17;3*5-3(6)1-2-4(7)8/h2*9-11,13-15H,6-8,12,16H2,1-5H3,(H,22,23);3*1-2H,(H,5,6)(H,7,8)
|
||||
| InChIKey |
AEOGOPZZLSQJKM-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | Muscarinic acetylcholine receptor M1 | Target Info | Agonist | [2] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| cAMP signaling pathway | |||||
| Neuroactive ligand-receptor interaction | |||||
| PI3K-Akt signaling pathway | |||||
| Cholinergic synapse | |||||
| Regulation of actin cytoskeleton | |||||
| PANTHER Pathway | Alzheimer disease-amyloid secretase pathway | ||||
| Heterotrimeric G-protein signaling pathway-Gq alpha and Go alpha mediated pathway | |||||
| Muscarinic acetylcholine receptor 1 and 3 signaling pathway | |||||
| Reactome | Muscarinic acetylcholine receptors | ||||
| G alpha (q) signalling events | |||||
| WikiPathways | Monoamine GPCRs | ||||
| Calcium Regulation in the Cardiac Cell | |||||
| Regulation of Actin Cytoskeleton | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| Secretion of Hydrochloric Acid in Parietal Cells | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800001599) | ||||
| REF 2 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

