Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0L0MB
|
||||
| Former ID |
DAP000844
|
||||
| Drug Name |
Acetophenazine
|
||||
| Synonyms |
Acephenazinum; Acetophenazina; Acetophenazinum; Tindal; Acetophenazine [INN]; Acetophenazina [INN-Spanish]; Acetophenazinum [INN-Latin]; Ketone, 10-(3-(4-(2-hydroxyethyl-1-piperazinyl)propyl)phenothiazin-2-yl methyl; 1-(10-{3-[4-(2-hydroxyethyl)piperazin-1-yl]propyl}-10H-phenothiazin-2-yl)ethanone; 1-[10-[3-[4-(2-hydroxyethyl)piperazin-1-yl]propyl]phenothiazin-2-yl]ethanone; 10-(3-(4-(2-Hydroxyethyl)-1-piperazinyl)propyl)phenothiazin-2-yl methyl ketone; 2-acetyl-10-[3-[4-(b-hydroxyethyl)piperazinyl]propyl]phenothiazine
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | False perceptions; Bipolar disorder [ICD9: 297, 780.1, 296.0, 296.1, 296.4, 296.5, 296.6, 296.7, 296.8, 300; ICD10:F22, R44, F31, F40-F42] | Approved | [1], [2] | ||
| Therapeutic Class |
Antipsychotic Agents
|
||||
| Structure |
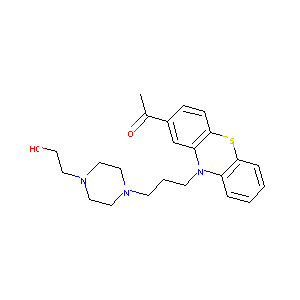
|
Download2D MOL |
|||
| Formula |
C23H29N3O2S
|
||||
| Canonical SMILES |
CC(=O)C1=CC2=C(C=C1)SC3=CC=CC=C3N2CCCN4CCN(CC4)CCO
|
||||
| InChI |
1S/C23H29N3O2S/c1-18(28)19-7-8-23-21(17-19)26(20-5-2-3-6-22(20)29-23)10-4-9-24-11-13-25(14-12-24)15-16-27/h2-3,5-8,17,27H,4,9-16H2,1H3
|
||||
| InChIKey |
WNTYBHLDCKXEOT-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 2751-68-0
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
9026, 6906047, 7978635, 8145915, 8163237, 14981427, 29285397, 46507036, 48415507, 50064639, 50125831, 57329848, 79129243, 85209527, 103301246, 104343061, 108136254, 117565968, 126671572, 127339546, 127339547, 129608156, 134982102, 135653585, 136350446, 137102211, 142677075, 160964397, 179116543, 184546003, 198962966, 223666661, 226471888, 250102248
|
||||
| ChEBI ID |
ChEBI:2401
|
||||
| SuperDrug ATC ID |
N05AB07
|
||||
| SuperDrug CAS ID |
cas=002751680
|
||||
| Target and Pathway | |||||
| Target(s) | D(2) dopamine receptor | Target Info | Antagonist | [3], [4] | |
| KEGG Pathway | Rap1 signaling pathway | ||||
| cAMP signaling pathway | |||||
| Neuroactive ligand-receptor interaction | |||||
| Gap junction | |||||
| Dopaminergic synapse | |||||
| Parkinson's disease | |||||
| Cocaine addiction | |||||
| Alcoholism | |||||
| PANTHER Pathway | Heterotrimeric G-protein signaling pathway-Gi alpha and Gs alpha mediated pathway | ||||
| Heterotrimeric G-protein signaling pathway-Gq alpha and Go alpha mediated pathway | |||||
| Dopamine receptor mediated signaling pathway | |||||
| Nicotine pharmacodynamics pathway | |||||
| Reactome | Dopamine receptors | ||||
| G alpha (i) signalling events | |||||
| WikiPathways | Hypothetical Network for Drug Addiction | ||||
| Monoamine GPCRs | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| Genes and (Common) Pathways Underlying Drug Addiction | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| Nicotine Activity on Dopaminergic Neurons | |||||
| References | |||||
| REF 1 | FDA Approved Drug Products from FDA Official Website. 2009. Application Number: (NDA) 012254. | ||||
| REF 2 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
| REF 3 | Atypical antipsychotics: mechanism of action. Can J Psychiatry. 2002 Feb;47(1):27-38. | ||||
| REF 4 | Specific therapeutic actions of acetophenazine, perphenazine, and benzquinamide in newly admitted schizophrenic patients. Clin Pharmacol Ther. 1967 Mar-Apr;8(2):249-55. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

