Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0K2ID
|
||||
| Former ID |
DIB013921
|
||||
| Drug Name |
NKTR-181
|
||||
| Indication | Pain [ICD9: 338, 356.0, 356.8,780; ICD10:G64, G90.0, R52, G89] | Phase 3 | [1] | ||
| Company |
Nektar therapeutics
|
||||
| Structure |
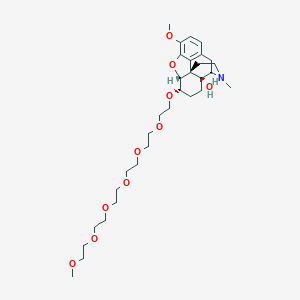
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | Mu-type opioid receptor | Target Info | Agonist | [2] | |
| KEGG Pathway | Neuroactive ligand-receptor interaction | ||||
| Estrogen signaling pathway | |||||
| Morphine addiction | |||||
| NetPath Pathway | TCR Signaling Pathway | ||||
| PANTHER Pathway | Heterotrimeric G-protein signaling pathway-Gi alpha and Gs alpha mediated pathway | ||||
| Heterotrimeric G-protein signaling pathway-Gq alpha and Go alpha mediated pathway | |||||
| Enkephalin release | |||||
| Pathway Interaction Database | IL4-mediated signaling events | ||||
| Reactome | Peptide ligand-binding receptors | ||||
| G alpha (i) signalling events | |||||
| WikiPathways | TCR Signaling Pathway | ||||
| GPCRs, Class A Rhodopsin-like | |||||
| Peptide GPCRs | |||||
| Opioid Signalling | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT02362672) Efficacy and Safety Study of NKTR-181 in Opioid-Naive Subjects With Low Back Pain. U.S. National Institutes of Health. | ||||
| REF 2 | A Review of Abuse-Deterrent Opioids For Chronic Nonmalignant Pain. P T. 2012 July; 37(7): 412-418. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

