Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0JY8T
|
||||
| Former ID |
DIB010764
|
||||
| Drug Name |
Bazedoxifene
|
||||
| Synonyms |
Brilence; Conbriza; Viviant; Bazedoxifene acetate; TSE-424; WAY-140424; WAY-TES 424
|
||||
| Drug Type |
Small molecular drug
|
||||
| Company |
Ligand Pharmaceuticals Inc
|
||||
| Structure |
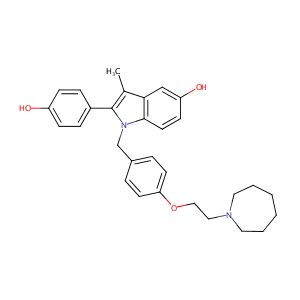
|
Download2D MOL |
|||
| Formula |
C30H34N2O3
|
||||
| InChI |
InChI=1S/C30H34N2O3/c1-22-28-20-26(34)12-15-29(28)32(30(22)24-8-10-25(33)11-9-24)21-23-6-13-27(14-7-23)35-19-18-31-16-4-2-3-5-17-31/h6-15,20,33-34H,2-5,16-19,21H2,1H3
|
||||
| InChIKey |
UCJGJABZCDBEDK-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | Estrogen receptor | Target Info | Modulator | [530223], [551871] | |
| Pathway Interaction Database | Regulation of nuclear SMAD2/3 signaling | ||||
| Signaling events mediated by HDAC Class II | |||||
| Plasma membrane estrogen receptor signaling | |||||
| LKB1 signaling events | |||||
| Regulation of Telomerase | |||||
| ATF-2 transcription factor network | |||||
| AP-1 transcription factor network | |||||
| FOXM1 transcription factor network | |||||
| Validated nuclear estrogen receptor alpha network | |||||
| Signaling mediated by p38-alpha and p38-beta | |||||
| FOXA1 transcription factor network | |||||
| References | |||||
| Ref 524681 | ClinicalTrials.gov (NCT02090400) Switching From Oral Bisphosphonates to Bazedoxifene to Evaluate Effects on Bone Mineral Density in Postmenopausal Women. U.S. National Institutes of Health. | ||||
| Ref 542376 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7355). | ||||
| Ref 551871 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
| Ref 530223 | Bazedoxifene, a selective estrogen receptor modulator: effects on the endometrium, ovaries, and breast from a randomized controlled trial in osteoporotic postmenopausal women. Menopause. 2009 Nov-Dec;16(6):1109-15. | ||||
| Ref 551871 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

