Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0F1WX
|
||||
| Former ID |
DNC001378
|
||||
| Drug Name |
SSR240600
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Urinary incontinence [ICD9: 788.3; ICD10:N39.3, N39.4, R32] | Phase 2 | [1] | ||
| Structure |
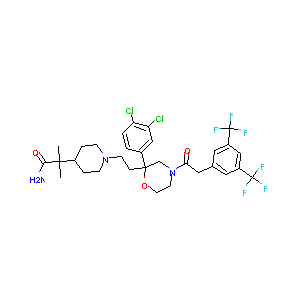
|
Download2D MOL |
|||
| Formula |
C31H35Cl2F6N3O3
|
||||
| Canonical SMILES |
CC(C)(C1CCN(CC1)CCC2(CN(CCO2)C(=O)CC3=CC(=CC(=C3)C(F)(F<br />)F)C(F)(F)F)C4=CC(=C(C=C4)Cl)Cl)C(=O)N
|
||||
| InChI |
1S/C31H35Cl2F6N3O3/c1-28(2,27(40)44)20-5-8-41(9-6-20)10-7-29(21-3-4-24(32)25(33)17-21)18-42(11-12-45-29)26(43)15-19-13-22(30(34,35)36)16-23(14-19)31(37,38)39/h3-4,13-14,16-17,20H,5-12,15,18H2,1-2H3,(H2,40,44)
|
||||
| InChIKey |
ZLNYUCXXSDDIFU-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 537034-22-3
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | Substance-P receptor | Target Info | Antagonist | [2] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Measles | |||||
| PANTHER Pathway | CCKR signaling map ST | ||||
| Reactome | G alpha (q) signalling events | ||||
| WikiPathways | SIDS Susceptibility Pathways | ||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| Spinal Cord Injury | |||||
| Peptide GPCRs | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT00174798) MILADY: A Randomized, Placebo-controlled Safety and Efficacy Trial of SSR240600C in Treatment of Overactive Bladder or Urge Urinary Incontinence.. U.S. National Institutes of Health. | ||||
| REF 2 | SSR240600 [(R)-2-(1-[2-[4-[2-[3,5-bis(trifluoromethyl)phenyl]acetyl]-2-(3,4-dichlorophenyl)-2-morpholinyl]ethyl]- 4-piperidinyl)-2-methylpropanamide], a centrally active nonpeptide antagonist of the tachykinin neurokinin-1 receptor: I. biochemical and pharmacological characterization. J Pharmacol Exp Ther. 2002 Dec;303(3):1171-9. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

