Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D09JFY
|
||||
| Former ID |
DCL000519
|
||||
| Drug Name |
Dutasteride + tamsulosin
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Prostate cancer; Benign prostatic hyperplasia [ICD9: 140-229, 185, 600; ICD10:C61, N40] | Phase 3 | [1] | ||
| Company |
GSK
|
||||
| Structure |
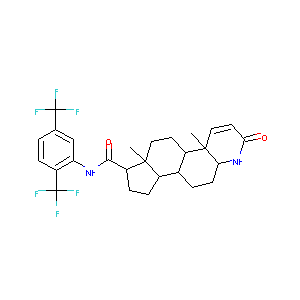
|
Download2D MOL |
|||
| Formula |
C47H58F6N4O7S
|
||||
| Canonical SMILES |
CCOC1=CC=CC=C1OCCNC(C)CC2=CC(=C(C=C2)OC)S(=O)(=O)N.CC12<br />CCC3C(C1CCC2C(=O)NC4=C(C=CC(=C4)C(F)(F)F)C(F)(F)F)CCC5C<br />3(C=CC(=O)N5)C
|
||||
| InChI |
1S/C27H30F6N2O2.C20H28N2O5S/c1-24-11-9-17-15(4-8-21-25(17,2)12-10-22(36)35-21)16(24)6-7-19(24)23(37)34-20-13-14(26(28,29)30)3-5-18(20)27(31,32)33;1-4-26-17-7-5-6-8-18(17)27-12-11-22-15(2)13-16-9-10-19(25-3)20(14-16)28(21,23)24/h3,5,10,12-13,15-17,19,21H,4,6-9,11H2,1-2H3,(H,34,37)(H,35,36);5-10,14-15,22H,4,11-13H2,1-3H3,(H2,21,23,24)/t15-,16-,17-,19+,21+,24-,25+;15-/m01/s1
|
||||
| InChIKey |
SHHCJPKACHSWFP-NMXGMQNUSA-N
|
||||
| CAS Number |
CAS 164656-23-9
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | Alpha-1A adrenergic receptor | Target Info | Antagonist | [2] | |
| 5-alpha reductase | Target Info | Inhibitor | [2] | ||
| KEGG Pathway | Calcium signaling pathway | ||||
| cGMP-PKG signaling pathway | |||||
| Neuroactive ligand-receptor interaction | |||||
| AMPK signaling pathway | |||||
| Adrenergic signaling in cardiomyocytes | |||||
| Vascular smooth muscle contraction | |||||
| Salivary secretion | |||||
| PANTHER Pathway | Alpha adrenergic receptor signaling pathway | ||||
| Reactome | Adrenoceptors | ||||
| G alpha (q) signalling events | |||||
| G alpha (12/13) signalling events | |||||
| WikiPathways | Monoamine GPCRs | ||||
| Calcium Regulation in the Cardiac Cell | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| Endothelin Pathways | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| AMPK Signaling | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT00090103) Benign Prostatic Hyperplasia Trial With Dutasteride And Tamsulosin Combination Treatment. U.S. National Institutes of Health. | ||||
| REF 2 | Clinical pipeline report, company report or official report of GlaxoSmithKline (2009). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

