Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D08HGY
|
||||
| Former ID |
DIB005659
|
||||
| Drug Name |
Naronapride
|
||||
| Synonyms |
Naronapride dihydrochloride; ATI-7500; ATI-7505; Cisapride analog, ARYx Therapeutics; ATI-7000 series, ARYx Therapeutics; Cisapride analog, Procter & Gamble; ATI-7000 series, Procter & Gamble; 5-HT 4 receptor agonist (oral, gastroesophageal reflux/irritable bowel syndrome/gastric motilitydisorder/constipation/dyspepsia), ARYx Therapeutics/Procter & Gamble Pharmaceuticals Inc
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Gastroesophageal reflux disease [ICD9: 140-229, 530; ICD10:K21] | Discontinued in Phase 2 | [1] | ||
| Company |
ARYx Therapeutics
|
||||
| Structure |
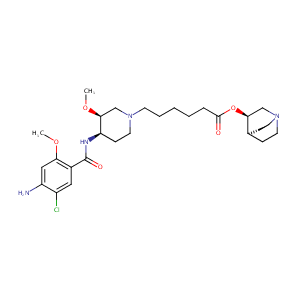
|
Download2D MOL |
|||
| Formula |
C27H41ClN4O5
|
||||
| Canonical SMILES |
c1(cc(c(cc1Cl)C(=O)N[C@H]1[C@H](CN(CC1)CCCCCC(=O)O[C@H]<br />1CN2CC[C@@H]1CC2)OC)OC)N
|
||||
| CAS Number |
CAS 860174-12-5
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | 5-hydroxytryptamine 4 receptor | Target Info | Agonist | [2] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| cAMP signaling pathway | |||||
| Neuroactive ligand-receptor interaction | |||||
| Serotonergic synapse | |||||
| PANTHER Pathway | Heterotrimeric G-protein signaling pathway-Gi alpha and Gs alpha mediated pathway | ||||
| 5HT4 type receptor mediated signaling pathway | |||||
| PathWhiz Pathway | Excitatory Neural Signalling Through 5-HTR 4 and Serotonin | ||||
| Reactome | Serotonin receptors | ||||
| G alpha (s) signalling events | |||||
| WikiPathways | Serotonin Receptor 4/6/7 and NR3C Signaling | ||||
| Monoamine GPCRs | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800019926) | ||||
| REF 2 | Systematic review: cardiovascular safety profile of 5-HT(4) agonists developed for gastrointestinal disorders. Aliment Pharmacol Ther. 2012 Apr;35(7):745-67. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

