Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D07NJI
|
||||
| Former ID |
DAP001450
|
||||
| Drug Name |
Treximet
|
||||
| Synonyms |
Trexima; Naproxen mixture with sumatriptan; Naproxen sodium mixture with sumatriptan succinate; Sumatriptan mixture with naproxen;Sumatriptan succinate and naproxen sodium; Sumatriptan succinate mixture with naproxen sodium; Naproxen / sumatriptan
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Migraine [ICD9: 346; ICD10:G43] | Approved | [1] | ||
| Company |
GSK
|
||||
| Structure |
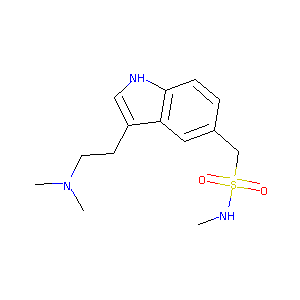
|
Download2D MOL |
|||
| Formula |
C32H40N3NaO9S
|
||||
| Canonical SMILES |
CC(C1=CC2=C(C=C1)C=C(C=C2)OC)C(=O)[O-].CNS(=O)(=O)CC1=C<br />C2=C(C=C1)NC=C2CCN(C)C.C(CC(=O)O)C(=O)O.[Na+]
|
||||
| InChI |
1S/C14H21N3O2S.C14H14O3.C4H6O4.Na/c1-15-20(18,19)10-11-4-5-14-13(8-11)12(9-16-14)6-7-17(2)3;1-9(14(15)16)10-3-4-12-8-13(17-2)6-5-11(12)7-10;5-3(6)1-2-4(7)8;/h4-5,8-9,15-16H,6-7,10H2,1-3H3;3-9H,1-2H3,(H,15,16);1-2H2,(H,5,6)(H,7,8);/q;;;+1/p-1/t;9-;;/m.0../s1
|
||||
| InChIKey |
IGRAQVLKCQVBQA-DBQHITQZSA-M
|
||||
| CAS Number |
CAS 154189-24-9
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | 5-hydroxytryptamine 1A receptor | Target Info | Antagonist | [2] | |
| KEGG Pathway | cAMP signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Serotonergic synapse | |||||
| PANTHER Pathway | Heterotrimeric G-protein signaling pathway-Gi alpha and Gs alpha mediated pathway | ||||
| 5HT1 type receptor mediated signaling pathway | |||||
| Reactome | Serotonin receptors | ||||
| G alpha (i) signalling events | |||||
| WikiPathways | Serotonin HTR1 Group and FOS Pathway | ||||
| SIDS Susceptibility Pathways | |||||
| Monoamine GPCRs | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | FDA Approved Drug Products from FDA Official Website. 2009. Application Number: (NDA) 021926. | ||||
| REF 2 | Clinical pipeline report, company report or official report of GlaxoSmithKline (2009). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

