Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D05LMT
|
||||
| Former ID |
DNC005390
|
||||
| Drug Name |
2-Phenyl-cyclopropylamine hydrochloride
|
||||
| Indication | Discovery agent | Investigative | [527283] | ||
| Structure |
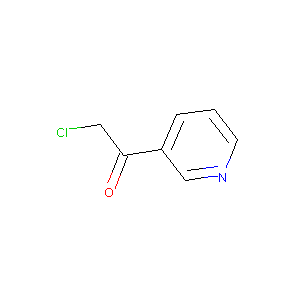
|
Download2D MOL |
|||
| Canonical SMILES |
C1=CC(=CN=C1)C(=O)CCl.Cl
|
||||
| InChI |
1S/C7H6ClNO.ClH/c8-4-7(10)6-2-1-3-9-5-6;/h1-3,5H,4H2;1H
|
||||
| InChIKey |
PBIUDEUWYGBHDW-UHFFFAOYSA-N
|
||||
| Target and Pathway | |||||
| Target(s) | Amine oxidase [flavin-containing] B | Target Info | Inhibitor | [527283] | |
| KEGG Pathway | Glycine, serine and threonine metabolism | ||||
| Arginine and proline metabolism | |||||
| Histidine metabolism | |||||
| Tyrosine metabolism | |||||
| Phenylalanine metabolism | |||||
| Tryptophan metabolism | |||||
| Drug metabolism - cytochrome P450 | |||||
| Metabolic pathways | |||||
| Serotonergic synapse | |||||
| Dopaminergic synapse | |||||
| Cocaine addiction | |||||
| Amphetamine addiction | |||||
| Alcoholism | |||||
| Pathway Interaction Database | Alpha-synuclein signaling | ||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

