Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D05GYL
|
||||
| Former ID |
DIB012706
|
||||
| Drug Name |
UR-63325
|
||||
| Synonyms |
DP-1; Histamine H4 antagonist (allergic rhinitis, asthma), Palau
|
||||
| Indication | Allergic rhinitis [ICD9: 472.0, 477, 995.3; ICD10:J00, J30, J31.0, T78.4] | Phase 2 | [1] | ||
| Company |
Palau Pharma SA
|
||||
| Structure |
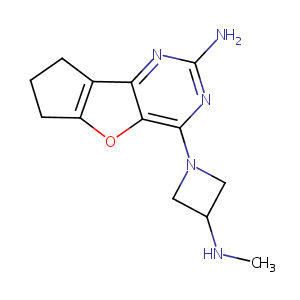
|
Download2D MOL |
|||
| Canonical SMILES |
c1(nc2c(c(n1)N1CC(C1)NC)oc1c2CCC1)N
|
||||
| Target and Pathway | |||||
| Target(s) | Histamine H4 receptor | Target Info | Antagonist | [2] | |
| KEGG Pathway | Neuroactive ligand-receptor interaction | ||||
| Reactome | Histamine receptors | ||||
| G alpha (i) signalling events | |||||
| WikiPathways | GPCR ligand binding | ||||
| GPCR downstream signaling | |||||
| GPCRs, Other | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT01260753) Proof of Activity Study of UR-63325 in Allergic Rhinitis Induced by Nasal Challenge. U.S. National Institutes of Health. | ||||
| REF 2 | Azines as histamine H4 receptor antagonists. Front Biosci (Schol Ed). 2012 Jan 1;4:967-87. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

