Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D04QSJ
|
||||
| Former ID |
DNC001188
|
||||
| Drug Name |
R0-93877
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Constipation [ICD9: 564; ICD10:K59.0] | Approved | [1], [2] | ||
| Structure |
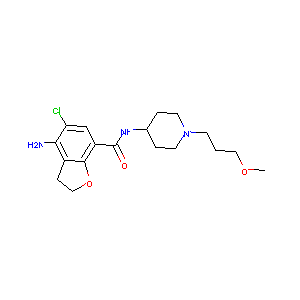
|
Download2D MOL |
|||
| Formula |
C18H26ClN3O3
|
||||
| Canonical SMILES |
COCCCN1CCC(CC1)NC(=O)C2=CC(=C(C3=C2OCC3)N)Cl
|
||||
| InChI |
1S/C18H26ClN3O3/c1-24-9-2-6-22-7-3-12(4-8-22)21-18(23)14-11-15(19)16(20)13-5-10-25-17(13)14/h11-12H,2-10,20H2,1H3,(H,21,23)
|
||||
| InChIKey |
ZPMNHBXQOOVQJL-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 86639-52-3
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
10056110, 14803834, 36118680, 50329889, 57353981, 85209596, 96025885, 103341576, 104015490, 111619007, 124772073, 126522466, 126666797, 126731460, 129936276, 134340386, 135131495, 135650861, 135697691, 136367812, 137009981, 142510835, 143497674, 152090515, 152159696, 152234885, 162009741, 162011596, 162205091, 164764612, 170466188, 170502890, 174006285, 174528011, 175438062, 179150084, 185997006, 188899543, 196408592, 198942073, 198993142, 210275056, 210280694, 223379010, 223662148, 226407081, 249734221, 251883665, 251971243, 252151346
|
||||
| Target and Pathway | |||||
| Target(s) | 5-hydroxytryptamine 4 receptor | Target Info | Agonist | [3] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| cAMP signaling pathway | |||||
| Neuroactive ligand-receptor interaction | |||||
| Serotonergic synapse | |||||
| PANTHER Pathway | Heterotrimeric G-protein signaling pathway-Gi alpha and Gs alpha mediated pathway | ||||
| 5HT4 type receptor mediated signaling pathway | |||||
| PathWhiz Pathway | Excitatory Neural Signalling Through 5-HTR 4 and Serotonin | ||||
| Reactome | Serotonin receptors | ||||
| G alpha (s) signalling events | |||||
| WikiPathways | Serotonin Receptor 4/6/7 and NR3C Signaling | ||||
| Monoamine GPCRs | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800009175) | ||||
| REF 2 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
| REF 3 | Irritable bowel syndrome: new agents targeting serotonin receptor subtypes. Drugs. 2001;61(3):317-32. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

