Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D04AEQ
|
||||
| Former ID |
DIB002281
|
||||
| Drug Name |
V81444
|
||||
| Indication | Parkinson's disease [ICD9: 332; ICD10:G20] | Phase 1/2 | [1] | ||
| Company |
Vernalis
|
||||
| Structure |
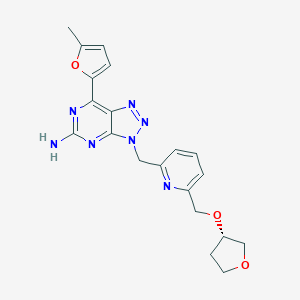
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | Adenosine A2a receptor | Target Info | Antagonist | [2] | |
| KEGG Pathway | Rap1 signaling pathway | ||||
| Calcium signaling pathway | |||||
| cAMP signaling pathway | |||||
| Neuroactive ligand-receptor interaction | |||||
| Vascular smooth muscle contraction | |||||
| Parkinson's disease | |||||
| Alcoholism | |||||
| Pathway Interaction Database | HIF-2-alpha transcription factor network | ||||
| PathWhiz Pathway | Intracellular Signalling Through Adenosine Receptor A2a and Adenosine | ||||
| Reactome | NGF-independant TRKA activation | ||||
| Adenosine P1 receptors | |||||
| G alpha (s) signalling events | |||||
| Surfactant metabolism | |||||
| WikiPathways | Nucleotide GPCRs | ||||
| Monoamine Transport | |||||
| GPCRs, Class A Rhodopsin-like | |||||
| NGF signalling via TRKA from the plasma membrane | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| GPCRs, Other | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT02253745) Safety, Tolerability, PK & Efficacy of V81444 in Volunteers With Attention Deficit/ Hyperactivity Disorder (ADHD). U.S. National Institutes of Health. | ||||
| REF 2 | Adenosine A2A receptor antagonists in Parkinson's disease: progress in clinical trials from the newly approved istradefylline to drugs in early development and those already discontinued. CNS Drugs. 2014 May;28(5):455-74. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

