Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D01AYX
|
||||
| Former ID |
DNCL003369
|
||||
| Drug Name |
AZD3293
|
||||
| Drug Type |
Small molecular drug
|
||||
| Company |
AstraZeneca
|
||||
| Structure |
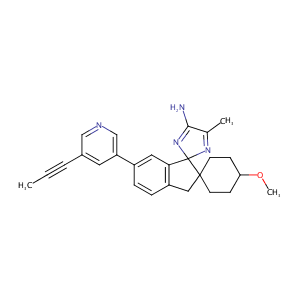
|
Download2D MOL |
|||
| Formula |
C26H28N4O
|
||||
| InChI |
InChI=1S/C26H28N4O/c1-4-5-18-12-21(16-28-15-18)19-6-7-20-14-25(10-8-22(31-3)9-11-25)26(23(20)13-19)29-17(2)24(27)30-26/h6-7,12-13,15-16,22H,8-11,14H2,1-3H3,(H2,27,30)
|
||||
| InChIKey |
WKDNQONLGXOZRG-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | Beta-secretase | Target Info | Modulator | [551416] | |
| References | |||||
| Ref 524922 | ClinicalTrials.gov (NCT02245737) An Efficacy and Safety Study of AZD3293 in Early Alzheimer's Disease. U.S. National Institutes of Health. | ||||
| Ref 542739 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7789). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

