Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0L6KC
|
||||
| Former ID |
DIB013164
|
||||
| Drug Name |
BILR-355
|
||||
| Synonyms |
BILR-355-BS; 11-Ethyl-5-methyl-8-[2-(1-oxidoquinolin-4-yloxy)ethyl]-6,11-dihydro-5H-dipyrido[3,2-b:2',3'-e][1,4]diazepin-6-one
|
||||
| Indication | Human immunodeficiency virus infection [ICD9: 279.3; ICD10:B20-B26] | Phase 2 | [521806] | ||
| Structure |
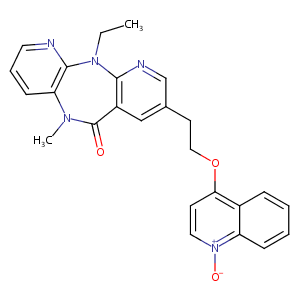
|
Download2D MOL |
|||
| Canonical SMILES |
CCN1c2ncccc2N(C)C(=O)c3cc(CCOc4cc[n+]([O-])c5ccccc45)cn<br />c13
|
||||
| InChI |
1S/C25H23N5O3/c1-3-29-23-19(25(31)28(2)21-9-6-12-26-24(21)29)15-17(16-27-23)11-14-33-22-10-13-30(32)20-8-5-4-7-18(20)22/h4-10,12-13,15-16H,3,11,14H2,1-2H3
|
||||
| InChIKey |
BEMBRAMZGVDPMH-UHFFFAOYSA-N
|
||||
| Target and Pathway | |||||
| Target(s) | HIV reverse transcriptase | Target Info | Inhibitor | [531281], [551871] | |
| References | |||||
| Ref 531281 | Evaluation of steady-state pharmacokinetic interactions between ritonavir-boosted BILR 355, a non-nucleoside reverse transcriptase inhibitor, and lamivudine/zidovudine in healthy subjects. J Clin Pharm Ther. 2012 Feb;37(1):81-8. | ||||
| Ref 551871 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

