Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0UI1P
|
||||
| Former ID |
DNC014165
|
||||
| Drug Name |
2-Cinnamamido-N4-hexyl-N1-hydroxysuccinamide
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Discovery agent | Investigative | [530409] | ||
| Structure |
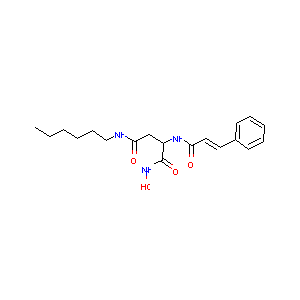
|
Download2D MOL |
|||
| Formula |
C19H27N3O4
|
||||
| Canonical SMILES |
CCCCCCNC(=O)CC(C(=O)NO)NC(=O)C=CC1=CC=CC=C1
|
||||
| InChI |
1S/C19H27N3O4/c1-2-3-4-8-13-20-18(24)14-16(19(25)22-26)21-17(23)12-11-15-9-6-5-7-10-15/h5-7,9-12,16,26H,2-4,8,13-14H2,1H3,(H,20,24)(H,21,23)(H,22,25)/b12-11+/t16-/m1/s1
|
||||
| InChIKey |
AOIMNHYQILMKAB-LPQFERQCSA-N
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Aminopeptidase N | Target Info | Inhibitor | [530409] | |
| BioCyc Pathway | Glutathione-mediated detoxification | ||||
| Pathway Interaction Database | C-MYB transcription factor network | ||||
| PathWhiz Pathway | Glutathione Metabolism | ||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

