Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0B8WN
|
||||
| Former ID |
DAP000340
|
||||
| Drug Name |
Ranitidine
|
||||
| Synonyms |
Achedos; Acidex; Alquen; Atural; Axoban; Biotidin; Coralen; Curan; Duractin; Ezopta; Fendibina; Gastrial; Gastridina; Gastrolav; Gastrosedol; Istomar; Kuracid; Logast; Mauran; Melfax; Microtid; Noctone; Ptinolin; Quantor; Quicran; RND; Radinat; Randin; Raniben; Raniberl; Ranibloc; Ranidine; Ranifur; Ranin; Raniogas; Raniplex; Ranisen; Raniter; Ranitidin; Ranitidina; Ranitidinum; Ranitiget; Ranitin; Rantacid; Rantidine; Ratic; Raticina; Sampep; Sostril; Taural; Terposen; Ulceranin; Ulcex; Ultidine; Urantac; Verlost; Vesyca; Vizerul; Weichilin; Weidos; Xanidine; ZANTAC; Zantab; Zantadin; Zantic; Ranitidine HCL; Rantidine HCL; Nu-Ranit; Rani-Q; Rani-nerton; Ranitidina [INN-Spanish]; Ranitidine (TN); Ranitidinum [INN-Latin]; Ul-Pep; Zantac (TN); Ranitidine (USAN/INN); Ranitidine [USAN:BAN:INN]; N-(2-((5-((Dimethylamino)methyl)furfuryl)thio)ethyl)-N'-methyl-2-nitro-1,1-ethenediamine; N (2-(((5-((Dimethylamino)methyl)-2-furanyl)methyl)thio)ethyl)-N'-methyl-2-nitro-1,1-ethenediamine; (E)-1-N'-[2-[[5-(dimethylaminomethyl)furan-2-yl]methylsulfanyl]ethyl]-1-N-methyl-2-nitroethene-1,1-diamine; (E)-N-{2-[({5-[(dimethylamino)methyl]-2-furyl}methyl)sulfanyl]ethyl}-N'-methyl-2-nitroethene-1,1-diamine; (E)-N-{2-[({5-[(dimethylamino)methyl]furan-2-yl}methyl)sulfanyl]ethyl}-N'-methyl-2-nitroethene-1,1-diamine
|
||||
| Drug Type |
Small molecular drug
|
||||
| Therapeutic Class |
Antiulcer Agents
|
||||
| Company |
GlaxoSmithKline
|
||||
| Structure |
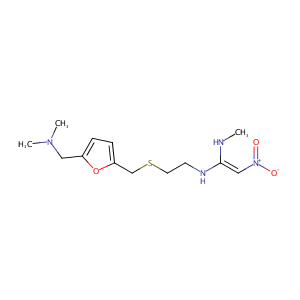
|
Download2D MOL |
|||
| Formula |
C13H22N4O3S
|
||||
| InChI |
InChI=1S/C13H22N4O3S/c1-14-13(9-17(18)19)15-6-7-21-10-12-5-4-11(20-12)8-16(2)3/h4-5,9,14-15H,6-8,10H2,1-3H3/b13-9+
|
||||
| InChIKey |
VMXUWOKSQNHOCA-UKTHLTGXSA-N
|
||||
| CAS Number |
CAS 66357-35-5
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
615112, 7847488, 7980484, 8149747, 10036397, 10532147, 11111714, 11111715, 11113371, 14776753, 26612202, 26680173, 26748519, 26748520, 26753732, 34666962, 46505543, 47216723, 47515261, 47810695, 48184944, 49846707, 49968693, 50100822, 50107423, 50107424, 50139267, 50286426, 53790194, 57410138, 85209253, 85245763, 90341455, 92124471, 92307665, 92711914, 93166337, 99301528, 103091649, 103155999, 103173566, 104171226, 111610678, 117377112, 117814891, 118258804, 124551883, 124636867, 124881290, 124881291
|
||||
| ChEBI ID |
ChEBI:8776
|
||||
| SuperDrug ATC ID |
A02BA02
|
||||
| SuperDrug CAS ID |
cas=066357355
|
||||
| Target and Pathway | |||||
| Target(s) | Histamine H2 receptor | Target Info | Antagonist | [535613], [535660], [538084] | |
| References | |||||
| Ref 537645 | Clinical review of histamine2 receptor antagonists. Arch Intern Med. 1990 Apr;150(4):745-51. | ||||
| Ref 538747 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 1234). | ||||
| Ref 535613 | Hemodynamic significance of histamine synthesis and histamine H1- and H2-receptor gene expression during endotoxemia. Naunyn Schmiedebergs Arch Pharmacol. 2002 Dec;366(6):513-21. Epub 2002 Oct 29. | ||||
| Ref 535660 | Knockouts model the 100 best-selling drugs--will they model the next 100? Nat Rev Drug Discov. 2003 Jan;2(1):38-51. | ||||
| Ref 538084 | Effect of the H2 histamine receptor antagonist on oxygen metabolism in some morphotic blood elements in patients with ulcer disease. Hepatogastroenterology. 1998 Jan-Feb;45(19):276-80. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

