Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0Y8ZN
|
||||
| Former ID |
DCL000582
|
||||
| Drug Name |
Ofatumumab
|
||||
| Synonyms |
Arzerra (TN)
|
||||
| Drug Type |
Antibody
|
||||
| Indication | Chronic lymphocytic leukaemia [ICD10:C91] | Approved | [1], [2] | ||
| Diffuse large B-cell lymphoma; Multiple sclerosis [ICD9: 202.8, 340; ICD10:C85.1, G35] | Phase 3 | [1], [2] | |||
| Rheumatold arthritis; Follicular lymphoma [ICD9: 202.0, 714; ICD10:C82, M05-M06] | Phase 2 | [1], [2] | |||
| Company |
GlaxoSmithKline
|
||||
| Structure |
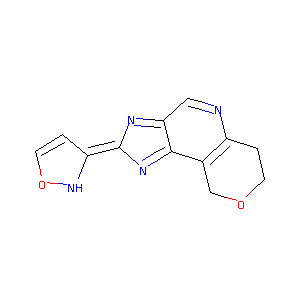
|
Download2D MOL |
|||
| Formula |
C12H10N4O2
|
||||
| PubChem Compound ID | |||||
| SuperDrug ATC ID |
L01XC10
|
||||
| Target and Pathway | |||||
| Target(s) | B-lymphocyte antigen CD20 | Target Info | [1], [3] | ||
| KEGG Pathway | Hematopoietic cell lineage | ||||
| References | |||||
| REF 1 | Hughes B: 2009 FDA drug approvals. Nat Rev Drug Discov. 2010 Feb;9(2):89-92. | ||||
| REF 2 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 6778). | ||||
| REF 3 | Clinical pipeline report, company report or official report of GlaxoSmithKline (2009). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

