| Drug General Information |
| Drug ID |
D0Q0EF
|
| Former ID |
DNCL003122
|
| Drug Name |
ITI-007
|
| Indication |
Schizophrenia; Sleep maintenance insomnia [ICD9: 295, 307.41, 307.42, 327.0, 780.51, 780.52; ICD10:F20, F51.0, G47.0]
|
Phase 3 |
[1],
[2]
|
|---|
| Company |
Intra-Cellular Therapies
|
| Structure |
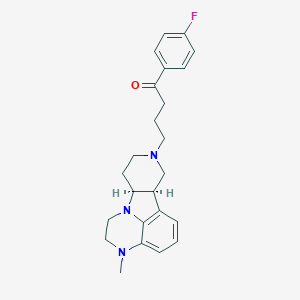
|
Download
2D MOL
3D MOL
|
| Target and Pathway |
| Target(s) |
Sodium-dependent serotonin transporter |
Target Info |
Antagonist |
[3]
|
|---|
| 5-hydroxytryptamine 2A receptor |
Target Info |
Antagonist |
[3]
|
|
KEGG Pathway
|
Serotonergic synapsehsa04020:Calcium signaling pathway
|
|
Neuroactive ligand-receptor interaction
|
|
Gap junction
|
|
Serotonergic synapse
|
|
Inflammatory mediator regulation of TRP channels
|
|
NetPath Pathway
|
TCR Signaling Pathway
|
|
PANTHER Pathway
|
5HT1 type receptor mediated signaling pathway
|
|
5HT2 type receptor mediated signaling pathway
|
|
5HT3 type receptor mediated signaling pathway
|
|
5HT4 type receptor mediated signaling pathwayP04374:5HT2 type receptor mediated signaling pathway
|
|
Reactome
|
Serotonin receptors
|
|
G alpha (q) signalling events
|
|
WikiPathways
|
Monoamine Transport
|
|
SIDS Susceptibility Pathways
|
|
NRF2 pathway
|
|
Synaptic Vesicle Pathway
|
|
Serotonin Transporter ActivityWP733:Serotonin Receptor 2 and STAT3 Signaling
|
|
Serotonin Receptor 2 and ELK-SRF/GATA4 signaling
|
|
Monoamine GPCRs
|
|
GPCRs, Class A Rhodopsin-like
|
|
Gastrin-CREB signalling pathway via PKC and MAPK
|
|
GPCR ligand binding
|
|
GPCR downstream signaling
|
|
GPCRs, Other
|
| References |
| REF 1 | ClinicalTrials.gov (NCT02469155) A Trial to Assess the Antipsychotic Efficacy of ITI-007 Over 6 Weeks of Treatment. |
|---|
| REF 2 | Clinical pipeline report, company report or official report of Intra-Cellular Therapies. |
|---|
| REF 3 | Clinical pipeline report, company report or official report of Intra-Cellular Therapies, Inc. |
|---|