Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0PM7Q
|
||||
| Former ID |
DCL000480
|
||||
| Drug Name |
AZD7762
|
||||
| Drug Type |
Small molecular drug
|
||||
| Company |
AstraZeneca
|
||||
| Structure |
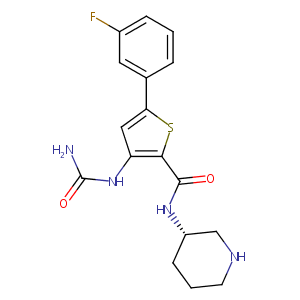
|
Download2D MOL |
|||
| Formula |
C17H19FN4O2S
|
||||
| InChI |
InChI=1S/C17H19FN4O2S/c18-11-4-1-3-10(7-11)14-8-13(22-17(19)24)15(25-14)16(23)21-12-5-2-6-20-9-12/h1,3-4,7-8,12,20H,2,5-6,9H2,(H,21,23)(H3,19,22,24)/t12-/m0/s1
|
||||
| InChIKey |
IAYGCINLNONXHY-LBPRGKRZSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
16231456, 23348024, 42207462, 75773863, 99437201, 117682512, 118847067, 124757322, 124950686, 125164126, 125329935, 131549273, 134213694, 135263746, 135727822, 136367458, 136368074, 136920283, 138468394, 144115659, 152258292, 152344041, 160647131, 160706048, 160868886, 162011707, 162037652, 163907978, 164045373, 170467355, 172912871, 174006971, 174561035, 186007043, 198971572, 203105583, 223366046, 224069960, 227368168, 244537251, 249582968, 252439982, 252450294
|
||||
| Target and Pathway | |||||
| Target(s) | Serine/threonine-protein kinase Chk1 | Target Info | Inhibitor | [550288] | |
| Pathway Interaction Database | Fanconi anemia pathway | ||||
| p73 transcription factor network | |||||
| ATR signaling pathway | |||||
| Circadian rhythm pathway | |||||
| p53 pathway | |||||
| Reactome | Activation of ATR in response to replication stress | ||||
| Processing of DNA double-strand break ends | |||||
| Presynaptic phase of homologous DNA pairing and strand exchange | |||||
| G2/M DNA damage checkpoint | |||||
| Ubiquitin Mediated Degradation of Phosphorylated Cdc25A | |||||
| Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex | |||||
| References | |||||
| Ref 522029 | ClinicalTrials.gov (NCT00473616) Phase I Single Ascending Dose/Multiple Ascending Dose in Patients Treated With AZD7762 and Irinotecan. U.S. National Institutes of Health. | ||||
| Ref 542688 | (http://www.guidetopharmacology.org/) Nucleic Acids Res. 2015 Oct 12. pii: gkv1037. The IUPHAR/BPS Guide to PHARMACOLOGY in 2016: towards curated quantitative interactions between 1300 protein targets and 6000 ligands. (Ligand id: 7713). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

