Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0O8UB
|
||||
| Former ID |
DNCL001944
|
||||
| Drug Name |
RG7129
|
||||
| Indication | Alzheimer disease [ICD9: 331; ICD10:G30] | Phase 1 | [1] | ||
| Company |
Roche
|
||||
| Structure |
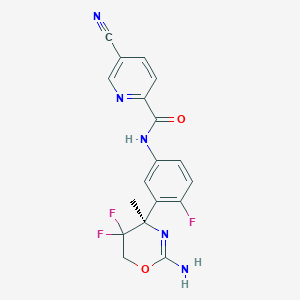
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | Beta-secretase | Target Info | Modulator | [2] | |
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT01592331) A Pharmacodynamic and Pharmacokinetic Study of RO5508887 in Healthy Volunteers. U.S. National Institutes of Health. | ||||
| REF 2 | BACE1 inhibitor drugs in clinical trials for Alzheimer's disease. Alzheimers Res Ther. 2014 Dec 24;6(9):89. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

