| Drug General Information |
| Drug ID |
D0NS1S
|
| Former ID |
DNC011126
|
| Drug Name |
ACACETIN
|
| Drug Type |
Small molecular drug
|
| Indication |
Discovery agent
|
Investigative |
[1]
|
|---|
| Structure |
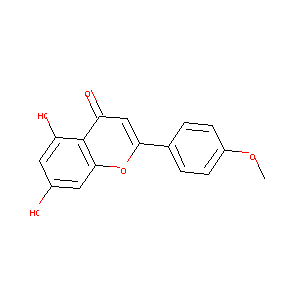
|
Download
2D MOL
3D MOL
|
| Formula |
C16H12O5
|
| Canonical SMILES |
COC1=CC=C(C=C1)C2=CC(=O)C3=C(C=C(C=C3O2)O)O
|
| InChI |
1S/C16H12O5/c1-20-11-4-2-9(3-5-11)14-8-13(19)16-12(18)6-10(17)7-15(16)21-14/h2-8,17-18H,1H3
|
| InChIKey |
DANYIYRPLHHOCZ-UHFFFAOYSA-N
|
| PubChem Compound ID |
|
| Target and Pathway |
| Target(s) |
Arachidonate 5-lipoxygenase |
Target Info |
Inhibitor |
[2]
|
|---|
| Cytochrome P450 1B1 |
Target Info |
Inhibitor |
[1]
|
| Xanthine dehydrogenase/oxidase |
Target Info |
Inhibitor |
[3]
|
|
BioCyc Pathway
|
Aspirin-triggered lipoxin biosynthesis
|
|
Resolvin D biosynthesis
|
|
Leukotriene biosynthesis
|
|
Lipoxin biosynthesis
|
|
Aspirin triggered resolvin D biosynthesis
|
|
Aspirin triggered resolvin E biosynthesisPWY66-401:Superpathway of tryptophan utilization
|
|
Superpathway of melatonin degradation
|
|
Melatonin degradation IPWY-6353:Purine nucleotides degradation
|
|
Urate biosynthesis/inosine 5'-phosphate degradation
|
|
Guanosine nucleotides degradation
|
|
Adenosine nucleotides degradation
|
|
Retinoate biosynthesis II
|
|
KEGG Pathway
|
Arachidonic acid metabolism
|
|
Metabolic pathways
|
|
Serotonergic synapse
|
|
Ovarian steroidogenesis
|
|
Toxoplasmosishsa00140:Steroid hormone biosynthesis
|
|
Tryptophan metabolism
|
|
Metabolism of xenobiotics by cytochrome P450
|
|
Chemical carcinogenesis
|
|
MicroRNAs in cancerhsa00230:Purine metabolism
|
|
Caffeine metabolism
|
|
Drug metabolism - other enzymes
|
|
Peroxisome
|
|
NetPath Pathway
|
IL4 Signaling PathwayNetPath_23:TSH Signaling Pathway
|
|
IL4 Signaling Pathway
|
|
TGF_beta_Receptor Signaling Pathway
|
|
PANTHER Pathway
|
Adenine and hypoxanthine salvage pathway
|
|
Purine metabolism
|
|
PathWhiz Pathway
|
Arachidonic Acid MetabolismPW000015:Caffeine Metabolism
|
|
Purine Metabolism
|
|
Reactome
|
Endogenous sterolsR-HSA-74259:Purine catabolism
|
|
WikiPathways
|
Vitamin D Receptor Pathway
|
|
Arachidonic acid metabolism
|
|
Eicosanoid Synthesis
|
|
Selenium Micronutrient NetworkWP702:Metapathway biotransformation
|
|
Estrogen metabolism
|
|
Benzo(a)pyrene metabolism
|
|
Tamoxifen metabolism
|
|
Tryptophan metabolism
|
|
Oxidation by Cytochrome P450
|
|
Nuclear Receptors Meta-Pathway
|
|
Estrogen Receptor Pathway
|
|
Sulindac Metabolic Pathway
|
|
Arylhydrocarbon receptor (AhR) signaling pathway
|
|
miR-targeted genes in muscle cell - TarBase
|
|
miR-targeted genes in lymphocytes - TarBase
|
|
miR-targeted genes in epithelium - TarBase
|
|
miR-targeted genes in adipocytes - TarBase
|
|
Phase 1 - Functionalization of compoundsWP408:Oxidative Stress
|
|
Effects of Nitric Oxide
|
|
Metabolism of nucleotides
|
|
Selenium Micronutrient Network
|
| References |
| REF 1 | Bioorg Med Chem. 2010 Sep 1;18(17):6310-5. Epub 2010 Jul 13.Selective inhibition of methoxyflavonoids on human CYP1B1 activity. |
|---|
| REF 2 | J Nat Prod. 1999 Sep;62(9):1241-5.Novel and known constituents from Buddleja species and their activity against leukocyte eicosanoid generation. |
|---|
| REF 3 | Inhibition of cow's milk xanthine oxidase by flavonoids. J Nat Prod. 1988 Mar-Apr;51(2):345-8. |
|---|