Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0N6SO
|
||||
| Former ID |
DCL000647
|
||||
| Drug Name |
SYR-472
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Metabolic disorders [ICD9: 270-279; ICD10:E70-E89] | Phase 3 | [1] | ||
| Company |
Takeda
|
||||
| Structure |
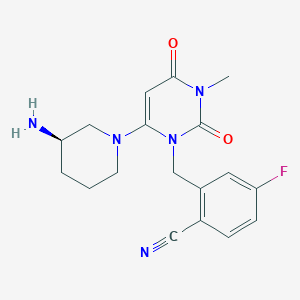
|
Download2D MOL |
|||
| Formula |
C22H26FN5O6
|
||||
| Canonical SMILES |
CN1C(=O)C=C(N(C1=O)CC2=C(C=CC(=C2)F)C#N)N3CCCC(C3)N.C(C<br />C(=O)O)C(=O)O
|
||||
| InChI |
1S/C18H20FN5O2.C4H6O4/c1-22-17(25)8-16(23-6-2-3-15(21)11-23)24(18(22)26)10-13-7-14(19)5-4-12(13)9-20;5-3(6)1-2-4(7)8/h4-5,7-8,15H,2-3,6,10-11,21H2,1H3;1-2H2,(H,5,6)(H,7,8)/t15-;/m1./s1
|
||||
| InChIKey |
OGCNTTUPLQTBJI-XFULWGLBSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| SuperDrug ATC ID |
R03AC09
|
||||
| SuperDrug CAS ID |
cas=018559596
|
||||
| Target and Pathway | |||||
| Target(s) | Dipeptidyl peptidase IV | Target Info | Inhibitor | [2], [3] | |
| KEGG Pathway | Protein digestion and absorption | ||||
| NetPath Pathway | IL2 Signaling Pathway | ||||
| TGF_beta_Receptor Signaling Pathway | |||||
| References | |||||
| REF 1 | Clinical pipeline report, company report or official report of Takeda. | ||||
| REF 2 | SYR-472, a novel once-weekly dipeptidyl peptidase-4 (DPP-4) inhibitor, in type 2 diabetes mellitus: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2014 Feb;2(2):125-32. | ||||
| REF 3 | Clinical pipeline report, company report or official report of Takeda (2009). | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

