Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D0B4JQ
|
||||
| Former ID |
DAP000328
|
||||
| Drug Name |
Tripelennamine
|
||||
| Synonyms |
Benzoxale; Cizaron; Dehistin; Piribenzil; Pyribenzamin; Pyribenzamine; Pyridbenzamine; Resistamine; Tonaril; Tripelannamine; Tripelenamina; Tripelenamine; Tripelennamin; Tripelennamina; Tripelennaminum; Tripellenamine; Triplennamine; Vetibenzamina; Pyrinamine base; Tripelennamina [Italian]; Tripelennamine [BAN]; Tripelennamine [INN]; Ts and Blues; Pyribenzamine (TN); Pyristine (piristina); Tripelenamina [INN-Spanish]; Tripelennamine (INN); Tripelennaminum [INN-Latin]; Tripelennamine Citrate (1:1); Beta-Dimethylaminoethyl-2-pyridylaminotoluene; Beta-Dimethylaminoethyl-2-pyridylbenzylamine; Benzyl-(alpha-pyridyl)-dimethylaethylendiamin; Benzyl-(alpha-pyridyl)-dimethylaethylendiamin [German]; Benzyl-(alpha.-pyridyl)-dimethylaethylendiamin; Benzyl-.alpha.-pyridyl-dimethyl-aethylendiamin; N-benzyl-N-[2-(dimethylamino)ethyl]pyridin-2-amine; N-Benzyl-N',N'-dimethyl-N-2-pyridylethylenediamine; N,N-Dimethyl-N'-benzyl-N'-(2-pyridyl)ethylenediamine; N,N-Dimethyl-N'-benzyl-N'-(alpha-pyridyl)ethylenediamine; N-Benzyl-N',N'-dimethyl-N-(2-pyridyl)ethylenediamine; N-Benzyl-N',N'-dimethyl-N-2-(pyridylethylene)diamine; N-Benzyl-N-(2-pyridyl)-N',N'-dimethyl ethylenediamine; N,N-Dimethyl-N'-benzyl-N'-(alpha.-pyridyl)ethylenediamine; N'-benzyl-N,N-dimethyl-N'-pyridin-2-ylethane-1,2-diamine; N,N-Dimethyl-N'-(phenylmethyl)-N'-2-pyridinyl-1,2-ethanediamine; N,N-dimethyl-N'-(phenylmethyl)-N'-pyridin-2-ylethane-1,2-diamine; N-Benzyl-N',N'-Dimethyl-N-Pyridin-2-Yl-Ethane-1,2-Diamine Hydrochloride; 1,2-Ethanediamine, N,N-dimethyl-N'-(phenylmethyl)-N'-2-pyridinyl-(9CI); 2-(Benzyl(2-dimethylaminoethyl)amino)pyridine; 2-(N-Benzyl-N-(2-dimethylaminoethyl)amino)pyridine; 2-(benzyl(2-(dimethylamino)ethyl)amino)pyridine; 2-[Benzyl(2-dimethylaminoethyl)amino]pyridine; 2-[N-Benzyl-N-(2-dimethylaminoethyl)amino]pyridine
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Hypersensitivity reactions; Coughs; Common colds [ICD10:R05] | Approved | [1] | ||
| Therapeutic Class |
Antiallergic Agents
|
||||
| Company |
Norvatis Phamaceuticals Corporation
|
||||
| Structure |
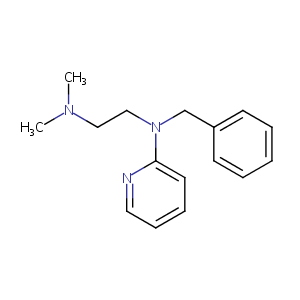
|
Download2D MOL |
|||
| Formula |
C16H21N3
|
||||
| InChI |
InChI=1S/C16H21N3/c1-18(2)12-13-19(16-10-6-7-11-17-16)14-15-8-4-3-5-9-15/h3-11H,12-14H2,1-2H3
|
||||
| InChIKey |
UFLGIAIHIAPJJC-UHFFFAOYSA-N
|
||||
| CAS Number |
CAS 91-81-6
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
9389, 413912, 3249497, 4760541, 7849633, 7980838, 8153440, 10537854, 11335709, 11360948, 11363035, 11365597, 11368159, 11372571, 11374006, 11376321, 11461920, 11484690, 11488773, 11491511, 11492189, 11493955, 14847861, 29224625, 46506125, 47291099, 47515290, 48035078, 48035079, 48416676, 50031251, 50100839, 56459444, 57322859, 76841200, 85209336, 85787988, 96025328, 103378362, 104309662, 117474948, 117474949, 124883327, 124883328, 126408182, 131342045, 134337640, 134971723, 136102146, 137001509
|
||||
| SuperDrug ATC ID |
D04AA04; R06AC04
|
||||
| SuperDrug CAS ID |
cas=000091816
|
||||
| Target and Pathway | |||||
| Target(s) | Histamine H1 receptor | Target Info | Antagonist | [2], [3], [4], [5] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Inflammatory mediator regulation of TRP channels | |||||
| PANTHER Pathway | Histamine H1 receptor mediated signaling pathway | ||||
| Reactome | Histamine receptors | ||||
| G alpha (q) signalling events | |||||
| WikiPathways | Monoamine GPCRs | ||||
| GPCRs, Class A Rhodopsin-like | |||||
| IL-4 Signaling Pathway | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health & Human Services. 2015 | ||||
| REF 2 | Histamine H1 and H2 receptor antagonists accelerate skin barrier repair and prevent epidermal hyperplasia induced by barrier disruption in a dry environment. J Invest Dermatol. 2001 Feb;116(2):261-5. | ||||
| REF 3 | Prostaglandin E2 aggravates gastric mucosal injury induced by histamine in rats through EP1 receptors. Life Sci. 2003 Dec 19;74(5):629-41. | ||||
| REF 4 | Role of N-methyl-D-aspartate receptors in gastric mucosal blood flow induced by histamine. J Neurosci Res. 2004 Sep 1;77(5):730-8. | ||||
| REF 5 | Involvement of histamine H1 and H2 receptors in the regulation of STAT-1 phosphorylation: inverse agonism exhibited by the receptor antagonists. Int Immunopharmacol. 2005 Jul;5(7-8):1299-309. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

