Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D09YVU
|
||||
| Former ID |
DNC008055
|
||||
| Drug Name |
6-(3-aminopropyl)benzo[h]isoquinolin-1(2H)-one
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Discovery agent | Investigative | [529069] | ||
| Structure |
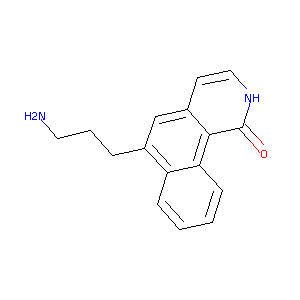
|
Download2D MOL |
|||
| Formula |
C16H16N2O
|
||||
| Canonical SMILES |
C1=CC=C2C(=C1)C(=CC3=C2C(=O)NC=C3)CCCN
|
||||
| InChI |
1S/C16H16N2O/c17-8-3-4-11-10-12-7-9-18-16(19)15(12)14-6-2-1-5-13(11)14/h1-2,5-7,9-10H,3-4,8,17H2,(H,18,19)
|
||||
| InChIKey |
LBNJYZFAUIDIPA-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| Target and Pathway | |||||
| Target(s) | Serine/threonine-protein kinase Chk1 | Target Info | Inhibitor | [529069] | |
| Pathway Interaction Database | Fanconi anemia pathway | ||||
| p73 transcription factor network | |||||
| ATR signaling pathway | |||||
| Circadian rhythm pathway | |||||
| p53 pathway | |||||
| Reactome | Activation of ATR in response to replication stress | ||||
| Processing of DNA double-strand break ends | |||||
| Presynaptic phase of homologous DNA pairing and strand exchange | |||||
| G2/M DNA damage checkpoint | |||||
| Ubiquitin Mediated Degradation of Phosphorylated Cdc25A | |||||
| Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex | |||||
| References | |||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

