Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D08HOM
|
||||
| Former ID |
DNCL002384
|
||||
| Drug Name |
ABT-348
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Hematological malignancies [ICD9: 200-209; ICD10:C81-C86] | Phase 2 | [1] | ||
| Company |
Abbott Laboratories Abbott
|
||||
| Structure |
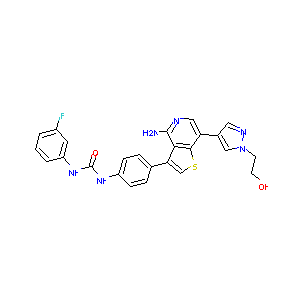
|
Download2D MOL |
|||
| Formula |
C25H21FN6O2S
|
||||
| Canonical SMILES |
C1=CC(=CC(=C1)F)NC(=O)NC2=CC=C(C=C2)C3=CSC4=C3C(=NC=C4C<br />5=CN(N=C5)CCO)N
|
||||
| InChI |
1S/C25H21FN6O2S/c26-17-2-1-3-19(10-17)31-25(34)30-18-6-4-15(5-7-18)21-14-35-23-20(12-28-24(27)22(21)23)16-11-29-32(13-16)8-9-33/h1-7,10-14,33H,8-9H2,(H2,27,28)(H2,30,31,34)
|
||||
| InChIKey |
WPHKIQPVPYJNAX-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID | |||||
| Target and Pathway | |||||
| Target(s) | Aurora protein kinase | Target Info | Modulator | [2] | |
| KEGG Pathway | Oocyte meiosis | ||||
| Reactome | APC/C:Cdh1 mediated degradation of Cdc20 and other APC/C:Cdh1 targeted proteins in late mitosis/early G1 | ||||
| Separation of Sister Chromatids | |||||
| Resolution of Sister Chromatid Cohesion | |||||
| RHO GTPases Activate Formins | |||||
| WikiPathways | EGF/EGFR Signaling Pathway | ||||
| JAK/STAT | |||||
| Gastric Cancer Network 1 | |||||
| Integrated Breast Cancer Pathway | |||||
| APC/C-mediated degradation of cell cycle proteins | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT02478320) Phase II Study of Ilorasertib (ABT348) in Patients With CDKN2A Deficient Solid Tumors. | ||||
| REF 2 | Preclinical characterization of ABT-348, a kinase inhibitor targeting the aurora, vascular endothelial growth factor receptor/platelet-derived growth factor receptor, and Src kinase families. J Pharmacol Exp Ther. 2012 Dec;343(3):617-27. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

