Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D06XWB
|
||||
| Former ID |
DNC000749
|
||||
| Drug Name |
Huperzine A
|
||||
| Drug Type |
Small molecular drug
|
||||
| Structure |
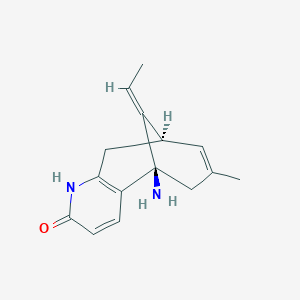
|
Download2D MOL |
|||
| Formula |
C15H18N2O
|
||||
| Canonical SMILES |
CC=C1C2CC3=C(C1(CC(=C2)C)N)C=CC(=O)N3
|
||||
| InChI |
1S/C15H18N2O/c1-3-11-10-6-9(2)8-15(11,16)12-4-5-14(18)17-13(12)7-10/h3-6,10H,7-8,16H2,1-2H3,(H,17,18)/b11-3+
|
||||
| InChIKey |
ZRJBHWIHUMBLCN-QDEBKDIKSA-N
|
||||
| CAS Number |
CAS 102518-79-6
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
24895708, 26756604, 29215281, 46511199, 46511231, 49991754, 49991755, 81041173, 85787773, 87462092, 91704460, 99301010, 99302530, 124892414, 134339656, 134339657, 134339658, 134340090, 135256104, 138769941, 142457861, 162093277, 162226717, 163395960, 163621066, 163686389, 163843547, 164758826, 180372184, 184548364, 216261296, 223435570, 226974040, 227296699, 242084598, 252455822
|
||||
| Target and Pathway | |||||
| Target(s) | Acetylcholinesterase | Target Info | Inhibitor | [535553] | |
| NMDA receptor | Target Info | Antagonist | [536934] | ||
| KEGG Pathway | Glycerophospholipid metabolism | ||||
| Cholinergic synapse | |||||
| Pathway Interaction Database | ATF-2 transcription factor network | ||||
| PathWhiz Pathway | Phospholipid Biosynthesis | ||||
| References | |||||
| Ref 535553 | Huperzine A attenuates cognitive deficits and brain injury in neonatal rats after hypoxia-ischemia. Brain Res. 2002 Sep 13;949(1-2):162-70. | ||||
| Ref 536934 | Progress report on new antiepileptic drugs: a summary of the Ninth Eilat Conference (EILAT IX). Epilepsy Res. 2009 Jan;83(1):1-43. Epub 2008 Nov 12. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

