Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D01JFM
|
||||
| Former ID |
DIB001402
|
||||
| Drug Name |
AC-170
|
||||
| Drug Type |
Small molecular drug
|
||||
| Indication | Allergic conjunctivitis [ICD9: 204.0, 372.0, 372.14, 995.3; ICD10:C91.0, H10, H10.45, T78.4] | Phase 3 | [1] | ||
| Company |
Aciex Therapeutics Inc
|
||||
| Structure |
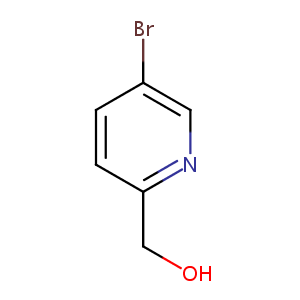
|
Download2D MOL |
|||
| Formula |
C6H6BrNO
|
||||
| Canonical SMILES |
C1=CC(=NC=C1Br)CO
|
||||
| InChI |
1S/C6H6BrNO/c7-5-1-2-6(4-9)8-3-5/h1-3,9H,4H2
|
||||
| InChIKey |
RUCZFWMEACWFER-UHFFFAOYSA-N
|
||||
| PubChem Compound ID | |||||
| PubChem Substance ID |
7734748, 8593186, 29203677, 39115928, 74318916, 75371566, 87561522, 87618057, 90271523, 92719515, 93579511, 99225392, 113775484, 118313039, 121360709, 124391777, 125359099, 126582968, 126590168, 126611515, 126620747, 126652955, 126671163, 126723742, 127629489, 129182084, 131391586, 131537930, 135721089, 136024226, 136370606, 136912257, 136984564, 136997320, 137269821, 140337842, 143438216, 143500659, 152244094, 152248436, 160806450, 160979238, 162147678, 162180243, 162298277, 162762909, 163810366, 164195039, 164757961, 170499800
|
||||
| Target and Pathway | |||||
| Target(s) | Histamine H1 receptor | Target Info | Antagonist | [2] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Inflammatory mediator regulation of TRP channels | |||||
| PANTHER Pathway | Histamine H1 receptor mediated signaling pathway | ||||
| Reactome | Histamine receptors | ||||
| G alpha (q) signalling events | |||||
| WikiPathways | Monoamine GPCRs | ||||
| GPCRs, Class A Rhodopsin-like | |||||
| IL-4 Signaling Pathway | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | ClinicalTrials.gov (NCT02132169) A Multi-Center Study Evaluating the Safety of AC-170 0.24%. U.S. National Institutes of Health. | ||||
| REF 2 | Clinical pipeline report, company report or official report of NicOx SA. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

