Drug Information
| Drug General Information | |||||
|---|---|---|---|---|---|
| Drug ID |
D00WJE
|
||||
| Former ID |
DIB009040
|
||||
| Drug Name |
SUN-1334H
|
||||
| Synonyms |
Histamine H1 receptor antagonist (oral, allergy) Sun Pharmaceutical
|
||||
| Indication | Allergic rhinitis [ICD9: 472.0, 477, 995.3; ICD10:J00, J30, J31.0, T78.4] | Discontinued in Phase 2 | [1] | ||
| Unspecified | Investigative | [2] | |||
| Company |
Sun Pharmaceutical Industries Ltd
|
||||
| Structure |
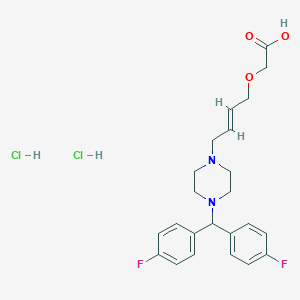
|
Download2D MOL |
|||
| Target and Pathway | |||||
| Target(s) | Histamine H1 receptor | Target Info | Antagonist | [2] | |
| KEGG Pathway | Calcium signaling pathway | ||||
| Neuroactive ligand-receptor interaction | |||||
| Inflammatory mediator regulation of TRP channels | |||||
| PANTHER Pathway | Histamine H1 receptor mediated signaling pathway | ||||
| Reactome | Histamine receptors | ||||
| G alpha (q) signalling events | |||||
| WikiPathways | Monoamine GPCRs | ||||
| GPCRs, Class A Rhodopsin-like | |||||
| IL-4 Signaling Pathway | |||||
| Gastrin-CREB signalling pathway via PKC and MAPK | |||||
| GPCR ligand binding | |||||
| GPCR downstream signaling | |||||
| References | |||||
| REF 1 | Trusted, scientifically sound profiles of drug programs, clinical trials, safety reports, and company deals, written by scientists. Springer. 2015. Adis Insight (drug id 800026836) | ||||
| REF 2 | Preclinical efficacy and safety pharmacology of SUN-1334H, a potent orally active antihistamine agent. Drugs R D. 2008;9(2):93-112. | ||||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

