Drug Information
| Drug General Information | Top | |||
|---|---|---|---|---|
| Drug ID |
D0OD2I
|
|||
| Drug Name |
Pralsetinib
|
|||
| Synonyms |
GBLBJPZSROAGMF-SIYOEGHHSA-N; Blu667; 2097132-94-8; SCHEMBL18806610; SCHEMBL18789229; SCHEMBL18789228; GTPL10033; BLU-668; EX-A1944; HY-112301; CS-0044766; N-[(1S)-1-[6-(4-fluoropyrazol-1-yl)pyridin-3-yl]ethyl]-1-methoxy-4-[4-methyl-6-[(5-methyl-1H-pyrazol-3-yl)amino]pyrimidin-2-yl]cyclohexane-1-carboxamide
Click to Show/Hide
|
|||
| Indication | Non-small-cell lung cancer [ICD-11: 2C25.Y; ICD-9: 162] | Approved | [1] | |
| Solid tumour/cancer [ICD-11: 2A00-2F9Z; ICD-10: C00-D48; ICD-9: 140-199, 210-229] | Phase 1 | [2] | ||
| Company |
Blueprint Medicines Cambridge, MA
|
|||
| Structure |
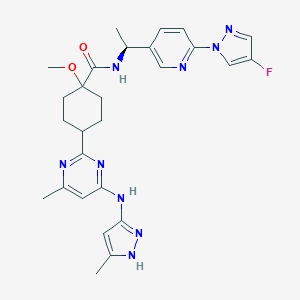 |
Download2D MOL |
||
| Formula |
C27H32FN9O2
|
|||
| Canonical SMILES |
CC1=CC(=NN1)NC2=NC(=NC(=C2)C)C3CCC(CC3)(C(=O)NC(C)C4=CN=C(C=C4)N5C=C(C=N5)F)OC
|
|||
| InChI |
1S/C27H32FN9O2/c1-16-11-22(33-23-12-17(2)35-36-23)34-25(31-16)19-7-9-27(39-4,10-8-19)26(38)32-18(3)20-5-6-24(29-13-20)37-15-21(28)14-30-37/h5-6,11-15,18-19H,7-10H2,1-4H3,(H,32,38)(H2,31,33,34,35,36)/t18-,19?,27?/m0/s1
|
|||
| InChIKey |
GBLBJPZSROAGMF-SIYOEGHHSA-N
|
|||
| CAS Number |
CAS 2097132-94-8
|
|||
| PubChem Compound ID | ||||
| Target and Pathway | Top | |||
|---|---|---|---|---|
| Target(s) | Proto-oncogene c-Ret (RET) | Target Info | Inhibitor | [1] |
| KEGG Pathway | Endocytosis | |||
| Pathways in cancer | ||||
| Thyroid cancer | ||||
| Central carbon metabolism in cancer | ||||
| Pathway Interaction Database | Signaling events regulated by Ret tyrosine kinase | |||
| Posttranslational regulation of adherens junction stability and dissassembly | ||||
| WikiPathways | SIDS Susceptibility Pathways | |||
| Dopaminergic Neurogenesis | ||||
| References | Top | |||
|---|---|---|---|---|
| REF 1 | Drugs@FDA. U.S. Food and Drug Administration. U.S. Department of Health Human Services. 2020 | |||
| REF 2 | Clinical pipeline report, company report or official report of the Pharmaceutical Research and Manufacturers of America (PhRMA) | |||
If You Find Any Error in Data or Bug in Web Service, Please Kindly Report It to Dr. Zhou and Dr. Zhang.

